
Abortion Providers Sue Mifepristone Restrictions
Abortion providers sue mifepristone restrictions—a headline that’s ignited a firestorm of debate across the nation. This legal battle isn’t just about a single drug; it’s about access to healthcare, women’s rights, and the very definition of reproductive freedom. The implications are vast, reaching far beyond courtrooms and impacting the lives of countless individuals. We’ll dive into the heart of this complex issue, exploring the legal arguments, the impact on access to care, and the broader social and political ramifications.
This case hinges on the FDA’s authority to regulate medications like mifepristone, a key component in medication abortion. Opponents argue the restrictions are necessary for safety, citing potential risks. Conversely, providers contend the restrictions are politically motivated and create unnecessary barriers to essential healthcare, disproportionately affecting vulnerable populations. The legal precedents cited, ranging from rulings on FDA authority to those concerning reproductive rights, form a complex web of arguments that are shaping the future of abortion access in America.
Legal Arguments in the Case
The legal battle surrounding the restrictions on Mifepristone, a medication used in medication abortion, centers on a complex interplay of FDA authority, drug safety regulations, and the right to access abortion care. Abortion providers challenged the restrictions imposed by various judges, arguing that they are not supported by scientific evidence and unduly burden access to a safe and effective medication.
The case hinges on the interpretation of existing laws and precedents related to drug approval and regulation, as well as the constitutional implications for reproductive healthcare access.
Core Legal Arguments of Abortion Providers
Abortion providers primarily argue that the FDA’s approval of Mifepristone, after rigorous review and decades of use, should be respected. They contend that the court’s interventions in the FDA’s regulatory process are unprecedented and undermine the agency’s scientific expertise. Their arguments emphasize the safety and efficacy of Mifepristone when used as directed, citing extensive data supporting its safe use.
They also argue that the restrictions on Mifepristone create unnecessary barriers to accessing abortion care, disproportionately impacting individuals in rural areas or those with limited financial resources. The core of their legal argument rests on the assertion that the restrictions violate the Administrative Procedure Act (APA), which governs how agencies like the FDA make decisions. They contend the court’s actions are arbitrary and capricious, lacking a rational basis in scientific evidence.
Legal Precedents Cited
Both sides in the case have cited numerous legal precedents. The abortion providers rely on cases affirming the FDA’s authority to regulate drugs and the importance of deference to agency expertise. They point to instances where courts have rejected challenges to FDA-approved drugs based on disagreements with the agency’s scientific judgment. Conversely, opponents of Mifepristone access have cited precedents that emphasize the court’s role in reviewing agency actions for compliance with statutory requirements.
They might point to cases where courts have found agency actions to be arbitrary or capricious, even if based on scientific evidence, if they fail to adequately address safety concerns. Specific case names and citations would be necessary for a complete analysis, but the general legal principles are as described.
FDA Authority and Mifepristone Safety
The arguments regarding FDA authority and Mifepristone’s safety are intrinsically linked. Abortion providers argue that the FDA’s rigorous approval process, including extensive clinical trials and ongoing monitoring, demonstrates the drug’s safety and efficacy. They contend that the restrictions are based on outdated information and disregard the overwhelming scientific consensus. Conversely, opponents argue that the FDA’s approval was flawed or that new evidence reveals risks not adequately considered during the initial approval.
They often cite anecdotal evidence or studies with questionable methodologies to support their claims, even while the overwhelming body of scientific evidence supports Mifepristone’s safety and efficacy. The core disagreement lies in the weight given to different sources of evidence and the interpretation of scientific data.
Potential Legal Consequences
The court’s decision will have far-reaching consequences for access to abortion care and the FDA’s regulatory authority. A ruling upholding the restrictions could significantly limit access to medication abortion, potentially forcing individuals to seek more invasive procedures or travel long distances for care. It could also set a precedent for future challenges to FDA-approved drugs, potentially undermining the agency’s ability to regulate medications effectively.
Conversely, a ruling striking down the restrictions would reinforce the FDA’s authority and reaffirm access to medication abortion. The potential impact extends beyond the immediate issue, affecting the balance of power between the judiciary and administrative agencies in regulating public health and safety. The Supreme Court’s decision inWest Virginia v. EPA* (2022) concerning the scope of agency authority provides a relevant parallel, illustrating the potential for significant changes to the regulatory landscape depending on the court’s interpretation of administrative law.
Impact on Access to Abortion
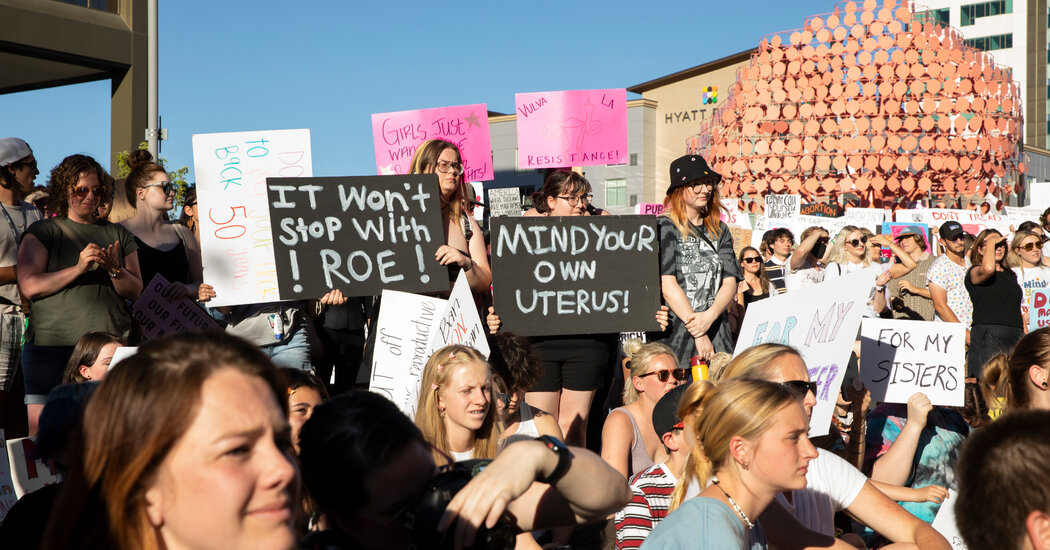
Source: nyt.com
The recent restrictions on mifepristone, a medication used in medication abortion, have created a ripple effect across the United States, significantly impacting access to abortion care and exacerbating existing inequalities in reproductive healthcare. The extent of this impact varies geographically, depending on pre-existing access to abortion services and the specific nature of state-level regulations.The restrictions on mifepristone will disproportionately affect women in rural areas and those with limited financial resources.
These women often face significant barriers to accessing healthcare, including long travel distances to clinics, lack of transportation, and inability to take time off work. The added hurdle of navigating more restrictive medication abortion protocols will further limit their options and potentially delay or prevent them from obtaining necessary care.
Geographic Variations in Access
The impact of the mifepristone restrictions is not uniform across the country. States with already restrictive abortion laws will experience a more dramatic decrease in access, while states with more permissive laws may see a less severe impact, though still noticeable. For instance, states with a high concentration of abortion clinics may still offer medication abortion, but the process will be more complex and potentially more expensive.
Conversely, states with few clinics or those that have already banned medication abortion will see almost complete elimination of this method, forcing individuals to seek out surgical abortions or travel long distances to states with more lenient regulations. This disparity in access will likely lead to increased health disparities between states and exacerbate existing inequalities.
Effects on Women’s Health and Reproductive Rights
Restricting access to mifepristone directly impacts women’s health and reproductive rights. Medication abortion is a safe and effective method preferred by many women due to its convenience and privacy. Limiting access to this method forces women to undergo surgical abortions, which carry a higher risk of complications. The increased difficulty in accessing abortion care may also lead to delayed care, resulting in more complex and potentially dangerous situations.
Furthermore, the restrictions infringe upon women’s autonomy over their bodies and their ability to make informed decisions about their reproductive health. The emotional toll of navigating increasingly restrictive legal and logistical barriers to abortion care should not be underestimated.
Alternative Methods of Abortion and Their Accessibility
While medication abortion using mifepristone is a common and preferred method, other abortion methods exist, including surgical abortion and the use of misoprostol alone. However, the accessibility and safety of these alternatives vary. Surgical abortion requires a clinic visit and may not be available in all areas, particularly in states with strict abortion regulations. Misoprostol alone can be used for medication abortion, but it is less effective than the combination of mifepristone and misoprostol and may lead to more complications.
The increased reliance on these alternative methods, due to mifepristone restrictions, could lead to an increase in complications and unsafe abortions, particularly for those lacking access to quality healthcare.
Hypothetical Scenario Illustrating Challenges
Imagine Sarah, a single mother working two minimum-wage jobs in rural Nebraska. She discovers she is pregnant and does not want to continue the pregnancy. Before the restrictions, she could have accessed medication abortion at a local clinic. Now, with the new restrictions, her options are severely limited. The nearest clinic offering surgical abortions is a six-hour drive away, requiring her to take time off work, potentially losing income and jeopardizing her job.
The cost of travel, accommodation, and the procedure itself is prohibitive. Without adequate financial resources or support, Sarah faces an impossible choice: continue an unwanted pregnancy or risk her financial stability and employment to seek care elsewhere. This scenario highlights the real-world challenges faced by women seeking abortion care under these new restrictions, particularly those in marginalized communities.
Political and Social Ramifications
The Supreme Court’s decision regarding mifepristone restrictions ignited a firestorm of political and social reactions, revealing deep divisions within American society on issues of reproductive rights, federal power, and the role of the judiciary. The ruling’s impact extended far beyond the legal realm, shaping political strategies, fueling social activism, and prompting significant shifts in public discourse.The political responses were sharply divided along partisan lines.
Republican politicians largely celebrated the decision, viewing it as a victory for their long-standing efforts to restrict abortion access. Many lauded the court’s perceived prioritization of fetal life and emphasized the need for greater protection for the unborn. Conversely, Democratic politicians and their allies strongly condemned the ruling, arguing it represented a dangerous overreach of judicial power and a significant setback for women’s health and reproductive freedom.
They framed the decision as an attack on bodily autonomy and access to essential healthcare. Various interest groups, such as Planned Parenthood and the National Right to Life Committee, also mobilized, using the ruling to further their respective agendas and galvanize their supporters.
Political Responses to the Court’s Decision
The political landscape following the ruling was characterized by intense polarization. Republican-controlled state legislatures in several states moved quickly to enact or strengthen existing abortion restrictions, while Democratic-controlled states worked to protect and expand access to abortion services. This led to a patchwork of laws across the country, creating significant disparities in abortion access based on geographic location.
The decision also fueled debates about the future of the Supreme Court and the appointment of justices, with both sides engaging in intense lobbying and advocacy efforts. For example, some states initiated legal challenges to the ruling, while others implemented measures to safeguard abortion providers and patients from potential legal repercussions.
Social and Cultural Debates Surrounding the Case
The case sparked intense social and cultural debates, moving beyond the purely political. Discussions revolved around the moral status of a fetus, the role of government in regulating healthcare decisions, and the implications for women’s equality. Religious beliefs played a significant role, with religious organizations and individuals holding strongly opposing views on the issue. These debates extended into the public sphere through protests, social media campaigns, and discussions in the media.
The restrictions on mifepristone also highlighted broader concerns about access to healthcare, particularly for women in underserved communities.
Comparison of Public Opinion on Abortion Access, Abortion providers sue mifepristone restrictions
Polling data reveals a shift in public opinion surrounding abortion access following the court’s decision. While consistent majorities have generally supported legal abortion access for some time, the specific nuances of public opinion varied depending on the circumstances of the abortion and the type of restrictions being considered. Before the ruling, polls often showed a significant portion of the public supporting abortion access in cases of rape, incest, or threat to the mother’s life.
Following the ruling, however, polls indicated increased concern about the potential impact on women’s health and reproductive freedom. This shift is reflected in the increased activism and engagement in political processes surrounding the issue.
The legal battles surrounding mifepristone restrictions are impacting abortion providers nationwide, forcing them to adapt and potentially impacting access to care. This comes at a time when, as reported by healthcare executives say talent acquisition labor shortages are a major business risk , the healthcare industry is already facing significant staffing shortages. These combined pressures could further limit abortion services and exacerbate existing healthcare inequalities.
Timeline of Key Events
A timeline illustrating the key events leading up to and following the lawsuit would include:
- [Date]: Initial lawsuit filed challenging the FDA’s approval of mifepristone.
- [Date]: Lower court rulings on the case.
- [Date]: Supreme Court hears the case.
- [Date]: Supreme Court issues its decision.
- [Date]: Immediate political and social reactions to the decision.
- [Date]: State-level legislative responses to the decision.
- [Date]: Ongoing legal challenges and activism surrounding the issue.
The Role of the FDA and Regulatory Processes

Source: yimg.com
The Food and Drug Administration (FDA) plays a crucial role in protecting public health by ensuring the safety and efficacy of medications, including mifepristone. Its regulatory process is complex, involving rigorous testing and review, and the recent legal challenges highlight the significant implications of its decisions on healthcare access and the future of drug regulation.The FDA’s authority stems from the Federal Food, Drug, and Cosmetic Act (FD&C Act), which empowers it to oversee the development, approval, and post-market surveillance of drugs.
This includes establishing standards for safety and efficacy, conducting reviews of clinical trial data, and monitoring adverse events after a drug is released to the market. For Mifepristone, the FDA’s role extends to its initial approval, subsequent risk evaluation and mitigation strategies (REMS), and ongoing monitoring of its safety profile.
Mifepristone’s FDA Approval Process
Mifepristone’s journey through the FDA’s approval process mirrored that of other drugs, albeit with heightened scrutiny given its role in medication abortion. The process began with pre-clinical studies, followed by extensive clinical trials designed to assess its safety and efficacy in inducing abortion. This data was then submitted to the FDA for review by expert panels, who analyzed the findings to determine whether the benefits of the drug outweighed its risks.
The FDA ultimately approved mifepristone under a Risk Evaluation and Mitigation Strategy (REMS), reflecting its acknowledgement of potential risks and the need for measures to mitigate them. This REMS included specific dispensing and prescribing guidelines. The approval process involved years of research, review, and debate.
Comparison to Other Medications
While the specifics of the regulatory pathway vary based on a drug’s mechanism of action and intended use, the FDA generally applies a consistent framework. Comparing mifepristone’s regulatory journey to other medications with similar safety profiles, such as certain cancer drugs with potentially serious side effects, reveals similarities in the required rigor of clinical trials and post-market surveillance. However, the political and social context surrounding mifepristone has undeniably influenced the level of public and political scrutiny.
The intense debate surrounding its use in abortion care has introduced a layer of complexity not typically seen with other medications, even those with comparable safety concerns.
Implications of Court Decisions on FDA Regulatory Processes
The court’s decision on the restrictions placed on mifepristone carries significant implications for the future of FDA regulatory processes. A ruling that significantly alters the FDA’s authority to approve and regulate drugs based on scientific evidence could set a precedent for future challenges to drug approvals. This could potentially lead to increased political influence in the drug approval process, potentially delaying or even preventing the approval of necessary medications, regardless of their safety and efficacy.
The potential chilling effect on the FDA’s ability to make independent scientific judgments could have far-reaching consequences for public health. For example, future medications with similar risk profiles to mifepristone, or even those addressing controversial conditions, might face heightened regulatory hurdles, potentially delaying access to life-saving treatments.
Alternative Medication and Treatment Options: Abortion Providers Sue Mifepristone Restrictions
The recent restrictions on mifepristone have sparked crucial conversations about alternative medications and treatment options for medication abortion. While mifepristone, in combination with misoprostol, is currently the most common and highly effective method, understanding the alternatives is vital for ensuring continued access to comprehensive reproductive healthcare. It’s important to note that the efficacy and safety of these alternatives vary, and access may be limited depending on location and individual circumstances.
The legal battles surrounding mifepristone restrictions are intense, with abortion providers fighting hard to maintain access. This fight highlights the complexities of healthcare access, especially considering the financial implications for large pharmacy chains like Walgreens, who recently raised their healthcare segment outlook following the Summit acquisition, as reported here: walgreens raises healthcare segment outlook summit acquisition. The Walgreens announcement underscores how significant changes in healthcare policy directly impact major players and ultimately, patient care, further emphasizing the stakes in the mifepristone case.
Alternative Medications for Medication Abortion
The availability and use of alternative medications for medication abortion are heavily dependent on the specific regulatory environment and the healthcare provider’s expertise. While mifepristol’s combination with misoprostol is the gold standard, misoprostol alone can induce abortion, though with potentially lower efficacy and higher risk of side effects. It’s crucial to consult a healthcare professional for accurate information and guidance.
| Medication Name | Description | Efficacy | Potential Side Effects |
|---|---|---|---|
| Misoprostol (Cytotec) | A prostaglandin analog; often used alone or in combination with mifepristone for medication abortion. | Effective, but generally less so than mifepristone/misoprostol combination. Efficacy varies depending on gestational age. | Cramping, bleeding, nausea, vomiting, diarrhea, fever, chills. Rarely, more serious side effects can occur. |
| Other Prostaglandins | Various prostaglandin analogs exist, though their use in medication abortion is less common than misoprostol. | Efficacy varies; generally less studied than misoprostol for this purpose. | Similar side effects to misoprostol, potentially with varying intensities. |
Efficacy and Safety Compared to Mifepristone
Studies consistently demonstrate the higher efficacy and safety of the mifepristone/misoprostol regimen compared to misoprostol alone. The combination reduces the likelihood of incomplete abortion and associated complications. Misoprostol used alone requires a higher dose, leading to a greater frequency and intensity of side effects. The use of alternative regimens should be guided by evidence-based guidelines and individual patient circumstances.
The legal battles surrounding abortion providers suing over mifepristone restrictions are intense, impacting healthcare access nationwide. These restrictions, and the resulting increased patient load on remaining providers, put a strain on already fragile systems, highlighting the importance of robust cybersecurity measures. This is especially crucial given the sensitive patient data involved, and the need for compliance with regulations like those detailed in the hhs healthcare cybersecurity framework hospital requirements cms framework.
The legal challenges to mifepristone only intensify the pressure on providers to maintain both patient care and data security.
The lack of mifepristone severely limits the ability to utilize the most effective and safest method.
Challenges in Accessing Alternative Options
Several factors can hinder access to alternative medication abortion options. These include: geographic limitations (fewer providers offering misoprostol-only regimens), regulatory restrictions on the prescribing or dispensing of misoprostol, the higher cost of treatment when only less effective options are available, and the potential for increased side effects requiring more medical intervention. Furthermore, a lack of comprehensive training on these alternative methods among healthcare providers can further restrict access.
Impact on Healthcare Providers
Restrictions on mifepristone significantly impact healthcare providers’ ability to offer comprehensive reproductive healthcare. The shift towards less effective and potentially riskier alternatives necessitates additional training, resources, and potentially increased patient monitoring. This places a greater burden on healthcare systems and individual providers, potentially affecting their capacity to provide other essential services. The need for more extensive counseling and post-procedure care increases the demand on resources and may lead to longer wait times and decreased access for patients.
Economic and Societal Costs
Restricting access to mifepristone carries significant economic and societal consequences, extending far beyond the immediate cost of the medication itself. These costs impact individuals, families, and the healthcare system as a whole, ultimately hindering societal well-being and economic productivity. Understanding these ramifications is crucial for a comprehensive assessment of the policy’s overall impact.The economic costs associated with restricting access to mifepristone are multifaceted.
Increased reliance on surgical abortions, a more expensive and invasive procedure, will place a greater financial burden on both individuals and the healthcare system. Travel costs, time off work, and childcare expenses associated with accessing surgical abortions in more distant locations will also escalate. Furthermore, the potential for complications from unsafe abortions resulting from restricted access to mifepristone will lead to increased healthcare costs related to treatment and emergency care.
These economic costs disproportionately affect low-income women and families, exacerbating existing inequalities.
Increased Healthcare Costs Due to Unsafe Abortions
Restricting access to mifepristone, a safe and effective medication, increases the likelihood of women resorting to unsafe abortion methods. These methods often result in serious complications, including severe infections, hemorrhage, incomplete abortions, and even death. The subsequent medical treatment required for these complications places a substantial strain on healthcare systems, increasing emergency room visits, hospitalizations, and long-term care needs.
The economic burden of managing these complications far surpasses the cost of providing safe and legal abortion care, including access to mifepristone. For instance, a single case of septic abortion requiring hospitalization and intensive care can cost tens of thousands of dollars, a cost that could have been avoided with access to safe abortion services.
Economic Burden on Women and Families
The economic impact on individual women and families is profound. The costs associated with travel to distant clinics, time off from work, childcare arrangements, and potential medical complications related to unsafe abortions can be financially devastating. Low-income women, who may already struggle to meet basic needs, are disproportionately affected. For example, a single mother working a minimum wage job might lose several days of income to travel for a surgical abortion, potentially leading to financial hardship and impacting her ability to provide for her family.
These economic pressures can have long-term consequences, including increased poverty and reduced opportunities for education and economic advancement.
Hypothetical Cost-Benefit Analysis: Mifepristone Access
A hypothetical cost-benefit analysis comparing the economic impacts of restricting versus maintaining access to mifepristone would likely demonstrate significant cost savings associated with maintaining access. The costs associated with unsafe abortions—including emergency medical care, long-term health complications, and lost productivity—would far outweigh the costs of providing safe and legal abortion services, including mifepristone. Furthermore, maintaining access to mifepristone allows women to manage their reproductive health more effectively, potentially reducing the need for more costly interventions later in life.
While precise figures are difficult to obtain due to the complexities of data collection and varying circumstances, several studies suggest that maintaining access to mifepristone is economically beneficial, both for individuals and society as a whole. A comprehensive analysis would require incorporating data on healthcare costs, lost productivity, and the long-term economic consequences of both restricted and unrestricted access to mifepristone.
Wrap-Up
The lawsuit challenging mifepristone restrictions is far more than a legal battle; it’s a reflection of deeply held beliefs about women’s health, bodily autonomy, and the role of government in personal decisions. The outcome will undeniably shape access to abortion care for years to come, impacting not only individual lives but also the broader political and social landscape. The ongoing discussion highlights the urgent need for open dialogue, respectful debate, and a commitment to ensuring equitable access to comprehensive reproductive healthcare for all.
Essential FAQs
What are the potential long-term effects of the court’s decision?
The long-term effects are uncertain but could include further restrictions on abortion access, impacting women’s health and reproductive freedom nationwide. It could also set precedents for future FDA regulations of other medications.
How might this affect women in rural areas?
Women in rural areas, who already face significant barriers to accessing healthcare, could be disproportionately affected by further restrictions on mifepristone, potentially leading to increased reliance on less safe methods of abortion.
What is the role of the pharmaceutical industry in this case?
The pharmaceutical industry has a vested interest in the outcome, as the decision could impact the regulation and marketing of other medications. Their involvement may manifest in amicus briefs or lobbying efforts.
Are there any international comparisons relevant to this case?
Yes, the legal arguments and access to medication abortion vary widely across countries, offering potential comparative analysis to inform the ongoing debate in the US.

