
340B Federal Clinics Pharma Drug Markups Explained
340B federal clinics pharma drug markups are a complex issue impacting healthcare affordability and access. This program, designed to help safety-net hospitals and clinics acquire medications at discounted prices, has sparked significant debate regarding pricing practices and the potential for inflated markups. We’ll delve into the intricacies of the 340B program, examining its history, the impact on federal clinics, and the ethical considerations surrounding drug pricing in this crucial area of healthcare.
From understanding the eligibility criteria and the discounts offered to analyzing the financial implications for participating clinics and the perspectives of pharmaceutical companies, we aim to provide a comprehensive overview. We’ll also explore the regulatory landscape, patient access, and the crucial role of transparency and accountability in ensuring responsible use of 340B savings. The goal is to shed light on this multifaceted issue and promote a more informed discussion.
340B Program Overview
The 340B Drug Pricing Program is a vital safety net for uninsured and underinsured patients, ensuring access to affordable medications. Established in 1992 as part of the Public Health Service Act, it allows covered entities to purchase outpatient drugs at significantly reduced prices, enabling them to stretch their resources and provide comprehensive care. This program plays a critical role in supporting safety-net hospitals and clinics serving vulnerable populations.The 340B program aims to improve access to prescription drugs for low-income patients by providing discounts to participating healthcare organizations.
These discounts allow covered entities to reinvest savings into patient care, expanding services and improving the overall quality of care for their communities. This program’s effectiveness is continuously evaluated and adjusted to meet the evolving needs of the healthcare landscape.
Eligibility Criteria for 340B Participation
Hospitals and clinics must meet specific criteria to participate in the 340B program. These criteria ensure that the program’s benefits reach the intended population of underserved patients. The process involves a rigorous application and verification process conducted by the Health Resources and Services Administration (HRSA). Key aspects of eligibility include serving a disproportionate share of low-income patients, demonstrating financial need, and meeting specific operational requirements.
Failure to meet these requirements can lead to the termination of 340B participation.
Types of Covered Entities Under the 340B Program
A variety of healthcare organizations qualify for 340B participation. The program encompasses a broad range of entities that provide care to underserved populations. These include:
- Disproportionate Share Hospitals (DSH): Hospitals that treat a significant number of low-income patients.
- Rural hospitals: Hospitals located in rural areas facing unique challenges in providing care.
- Community health centers: Federally qualified health centers serving medically underserved populations.
- Freestanding cancer hospitals: Hospitals specializing in cancer care.
- Children’s hospitals: Hospitals providing specialized care for children.
This diverse group of entities ensures that the program reaches a wide range of patients in need. Each entity undergoes a thorough review to confirm their eligibility based on specific criteria.
340B Drug Pricing Discounts
The 340B program offers significant discounts on the average manufacturer price (AMP) of covered outpatient drugs. The exact discount varies depending on the drug manufacturer and the specific drug, but it can be substantial, often representing a significant cost savings for participating entities. These savings are crucial for allowing these organizations to provide essential care to their communities.
For example, a drug with an AMP of $100 might be available to a 340B participant for $20 or less, depending on various factors. This substantial reduction allows these organizations to extend their services and improve the quality of care they can provide. These savings are not guaranteed to be consistent across all manufacturers and drugs, and pricing is subject to change.
Drug Markups and Pricing Practices
The 340B drug pricing program, while intended to help safety-net hospitals and clinics serve vulnerable populations, has become a complex issue due to the significant markups on drug prices. Understanding how pharmaceutical companies set prices and how those prices differ under 340B compared to the commercial market is crucial to evaluating the program’s effectiveness and its impact on both providers and patients.
The 340B drug pricing program for federal clinics is constantly under scrutiny, with debates raging over fair pricing and potential markups. This issue becomes even more complex when considering major pharmacy chains like Walgreens, who recently raised their healthcare segment outlook following the Summit acquisition, as seen in this article: walgreens raises healthcare segment outlook summit acquisition.
How this acquisition and increased profitability will affect their participation in the 340B program, and ultimately the cost of drugs for patients in these clinics, remains a key question.
This section delves into the intricacies of drug pricing and the financial implications of these markups for participating clinics.
Pharmaceutical companies employ a multifaceted approach to drug pricing. Factors considered often include research and development costs, manufacturing expenses, marketing and sales efforts, and the anticipated market demand. A crucial element is also the drug’s perceived value, considering its efficacy, safety profile, and potential to improve patient outcomes. The final price is then set to maximize profitability while remaining competitive within the market.
For example, a groundbreaking new cancer treatment might command a higher price due to its potential life-saving benefits, even if the manufacturing costs are relatively high. Conversely, a generic drug, lacking patent protection, would be priced significantly lower due to increased competition.
340B Drug Prices Compared to Commercial Market Prices
Drugs purchased through the 340B program are typically sold to participating clinics at a discounted price compared to the prices paid by commercial payers, such as private insurance companies. However, this discount is not uniform across all drugs and manufacturers. Some manufacturers offer substantial discounts, while others provide only minimal reductions. The extent of the discount often depends on the specific drug, the manufacturer’s pricing strategy, and the negotiating power of the 340B covered entity.
This variability leads to significant differences in the actual cost of medications obtained through the 340B program versus the commercial market. A hypothetical example: a drug priced at $100 in the commercial market might be available for $70 under the 340B program, representing a 30% discount. However, another drug might only see a 5% discount, highlighting the wide variation.
Factors Contributing to Price Discrepancies
Several factors contribute to the discrepancies observed between 340B and commercial drug prices. These include the aforementioned manufacturer pricing strategies, the volume of drugs purchased by 340B providers (larger volume purchases often lead to better discounts), and the overall market dynamics for a given drug. Additionally, the complexity of the 340B program itself, including contract negotiations and the various reporting requirements, can indirectly impact the pricing landscape.
For instance, the administrative burden associated with 340B participation might influence a manufacturer’s willingness to offer deeper discounts. Furthermore, the varying financial situations and bargaining power of different 340B covered entities could also lead to differing drug acquisition costs.
Financial Implications of Drug Markups for Participating Clinics
While 340B discounts are intended to help clinics reduce their drug costs, the practice of markups, where clinics resell drugs to other entities (sometimes at inflated prices), significantly impacts the program’s financial implications. The ability of clinics to generate revenue from these markups can be substantial and affects their overall financial health. However, this practice has also raised ethical and legal concerns, especially when markups exceed the costs incurred by the clinics.
For instance, a clinic acquiring a drug at a 30% discount under 340B might resell it at a price closer to the commercial rate, generating a profit margin significantly higher than their actual acquisition and administrative costs. This creates a financial incentive to participate in the 340B program and potentially to engage in practices that have been criticized as potentially undermining the program’s original intent.
Impact on Federal Clinics
The 340B Drug Pricing Program offers significant advantages to federal clinics, allowing them to stretch their budgets and enhance patient care. However, participation also presents unique challenges related to inventory management and program compliance. Understanding both the benefits and drawbacks is crucial for optimizing the program’s impact on these vital healthcare providers.
Benefits of 340B Participation for Federal Clinics
Federal clinics participating in the 340B program receive substantial discounts on outpatient drugs, leading to significant cost savings. These savings can be redirected towards improving various aspects of patient care, such as expanding services, upgrading equipment, or hiring additional staff. The increased purchasing power allows clinics to offer a wider range of medications and potentially improve patient access to necessary treatments.
This ultimately contributes to better health outcomes for the underserved populations these clinics typically serve.
Challenges in Managing 340B Drug Inventory and Distribution
Managing the 340B program effectively presents several logistical hurdles for federal clinics. Accurate tracking of 340B drugs, ensuring proper segregation from non-340B inventory, and maintaining meticulous record-keeping are essential for compliance. The complexities of the program’s regulations and the potential for audits add to the administrative burden. Furthermore, the need for robust inventory management systems to prevent drug waste or expiration is paramount.
Efficient distribution systems are also crucial to ensure timely access to medications for patients.
Examples of 340B Savings Utilization
Federal clinics have demonstrated diverse and impactful uses for their 340B savings. For instance, the Indian Health Service (IHS) has used these savings to improve access to specialty medications for chronic conditions like diabetes and hypertension in remote communities. Other clinics may invest in advanced diagnostic equipment, improving the accuracy and speed of diagnoses. Some clinics have used 340B savings to expand their behavioral health services, addressing the significant mental health needs of their patient populations.
Finally, investment in staff training and professional development programs enhances the quality of care provided.
Impact of 340B on Different Types of Federal Clinics
| Clinic Type | Average Savings | Challenges Faced | Utilization of Savings |
|---|---|---|---|
| Indian Health Service (IHS) Clinics | Varies significantly based on location and patient population; estimates suggest millions annually for larger facilities. | Logistical challenges in remote areas, limited staffing, and complex regulatory compliance. | Expanding access to specialty medications, upgrading telehealth infrastructure, improving primary care services. |
| Department of Veterans Affairs (VA) Clinics | Significant savings, though precise figures are not publicly released due to internal accounting practices. | Balancing 340B compliance with existing VA inventory and distribution systems. | Investing in new technologies, enhancing patient care programs, and supporting research initiatives. |
| Public Health Service (PHS) Clinics | Savings vary depending on the size and location of the clinic, often used to supplement limited budgets. | Maintaining accurate inventory tracking and ensuring compliance with complex regulations. | Improving access to preventative care, expanding health education programs, and upgrading facilities. |
| Military Treatment Facilities (MTFs) | Participation is less common due to existing purchasing power, though some may participate in specific programs. | Integrating 340B compliance with military procurement and supply chain systems. | Supplementing existing resources for specific patient populations or programs. |
Regulatory Landscape and Oversight
The 340B Drug Pricing Program, while designed to assist safety-net hospitals and clinics in serving vulnerable populations, operates within a complex regulatory environment. Understanding this framework is crucial to both ensuring program integrity and maximizing its benefits for participating providers. The Health Resources and Services Administration (HRSA) plays a central role in this oversight, shaping the program’s direction and enforcing compliance.The Health Resources and Services Administration (HRSA) is the primary federal agency responsible for overseeing the 340B program.
HRSA establishes the program’s regulations, monitors compliance among participating entities, and addresses issues of non-compliance. This involves interpreting the statutory requirements, issuing guidance documents, conducting audits, and enforcing penalties for violations. HRSA’s oversight aims to ensure that the program’s goals are met while preventing fraud, waste, and abuse. They achieve this through a multifaceted approach including regular reporting requirements from participating entities, on-site reviews, and data analysis to identify potential issues.
HRSA’s Role in 340B Program Oversight
HRSA’s responsibilities extend to several key areas. They develop and publish regulations clarifying the program’s requirements, including eligibility criteria, allowable drug pricing practices, and record-keeping standards. They also provide technical assistance and training to participating providers to help them understand and comply with these regulations. Furthermore, HRSA conducts audits and investigations to ensure that participating entities are properly utilizing the 340B discounts and adhering to the program’s rules.
Sanctions for non-compliance can range from warnings and corrective action plans to suspension or termination from the program. This comprehensive approach aims to balance supporting access to affordable medications with safeguarding the integrity of the program.
340B Program Regulations and Compliance Requirements
Participating entities must adhere to a detailed set of regulations to maintain their 340B program participation. These regulations cover various aspects of the program, including eligibility criteria, drug acquisition and dispensing practices, record-keeping requirements, and reporting obligations. For example, covered entities must demonstrate that they serve a disproportionate share of low-income patients and meet specific criteria related to their patient demographics and service areas.
Accurate record-keeping is paramount, including detailed documentation of drug acquisition costs, patient eligibility verification, and drug dispensing procedures. Failure to comply with these regulations can result in significant penalties, including fines and exclusion from the program. Regular self-assessments and internal audits are often recommended to proactively identify and address potential compliance issues.
Potential Areas for Improvement in the 340B Regulatory Framework
While the current regulatory framework provides a foundation for oversight, several areas could benefit from improvement. One key area is enhancing clarity and consistency in the regulations themselves. Ambiguities in the current rules can lead to inconsistent interpretations and potential non-compliance, even when entities are acting in good faith. Streamlining the reporting requirements could also reduce the administrative burden on participating entities, allowing them to focus more on patient care.
Finally, strengthening enforcement mechanisms could deter fraudulent activities and ensure accountability among all stakeholders. A more robust data-driven approach to oversight, incorporating advanced analytics and technology, could improve the efficiency and effectiveness of HRSA’s monitoring efforts.
340B Drug Acquisition and Dispensing Process
This flowchart illustrates the typical process of 340B drug acquisition and dispensing.[Imagine a flowchart here. The flowchart would begin with “Covered Entity Identifies Eligible Patient,” branching to “Verify Patient Eligibility through HRSA Data or Other Means,” then to “Submit Order to 340B Pharmaceutical Manufacturer,” followed by “Manufacturer Ships Drugs at 340B Price,” then to “Covered Entity Dispenses Drug to Eligible Patient,” and finally to “Maintain Accurate Records and Reporting to HRSA.”] The process highlights the key steps involved, from patient eligibility verification to drug dispensing and record-keeping, emphasizing the importance of accurate documentation at each stage to ensure compliance with 340B program regulations.
This clear visual representation simplifies the often complex procedures associated with 340B drug acquisition and dispensing.
Patient Access and Affordability
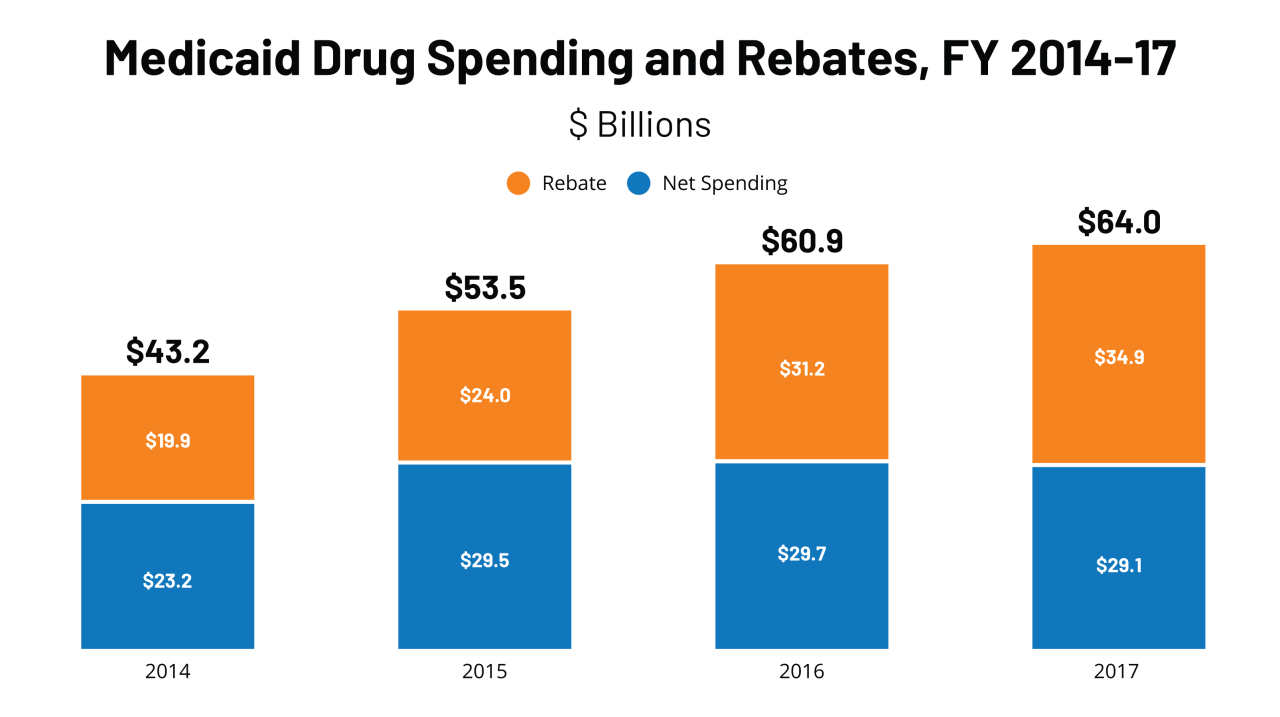
Source: kff.org
The 340B Drug Pricing Program aims to improve patient access to affordable medications for safety-net providers. However, the impact on actual patient access and affordability is a complex issue with varying perspectives and ongoing debate. Understanding this impact requires examining both the potential benefits and the potential drawbacks of the program.The core argument for 340B is that discounted drug pricing allows safety-net clinics to stretch their budgets, enabling them to offer more comprehensive care and potentially reach more patients.
This increased capacity could translate to improved access to essential medications for vulnerable populations who might otherwise struggle to afford them. However, concerns exist regarding whether these savings are consistently translated into improved patient access and lower out-of-pocket costs. Some argue that the increased revenue generated through 340B drug markups might not always directly benefit patients.
Patient Access to Medications
Studies examining the direct impact of 340B on patient access to medications have yielded mixed results. Some research suggests a positive correlation between 340B participation and increased medication dispensing rates, particularly for chronic conditions. These studies often point to the ability of clinics to offer more comprehensive medication management services due to the cost savings from the 340B program.
Conversely, other research has highlighted that the benefits are not always consistently observed across all participating clinics and patient populations. Factors like clinic operational efficiency and patient demographics significantly influence the extent to which 340B savings translate into increased medication access. For example, a clinic with efficient inventory management and robust patient outreach programs might demonstrate greater success in using 340B savings to improve patient access compared to a clinic lacking these capabilities.
Comparison of Patient Outcomes
Direct comparisons of patient outcomes between 340B participating clinics and non-participating clinics are challenging due to the complexities of controlling for confounding variables. Factors like patient demographics, the types of services offered, and the overall health status of the patient population can significantly influence health outcomes. However, some studies have attempted to address these complexities through statistical modeling and control groups.
While findings are not universally consistent, some research indicates that 340B participation might be associated with improved medication adherence rates and better management of chronic conditions in certain populations. This suggests a potential positive impact on overall patient health outcomes. Further research with more robust methodologies is needed to definitively establish a causal link between 340B participation and improved patient outcomes.
Potential for Improved Patient Care Through 340B Savings
The potential exists for 340B savings to significantly enhance patient care. These savings could be channeled towards various initiatives, including expanded access to specialized care, increased staffing levels, investment in new technologies, and enhanced patient education programs. For instance, a clinic might use 340B savings to hire additional nurses or social workers to improve patient support and follow-up care.
They might also invest in telehealth technologies to improve access to specialists for patients in remote areas. The key is ensuring that the program’s financial benefits are strategically invested to directly improve the quality and accessibility of patient care.
Strategies to Maximize Patient Benefits from the 340B Program, 340b federal clinics pharma drug markups
Effective utilization of 340B savings to maximize patient benefits requires careful planning and strategic implementation.
- Transparent Financial Management: Establishing clear financial tracking systems to monitor how 340B savings are used and ensure transparency in resource allocation.
- Targeted Investments in Patient Care: Prioritizing investments in programs and services that directly improve patient access to medications and other essential healthcare services.
- Enhanced Patient Outreach and Education: Implementing proactive strategies to ensure patients understand the benefits of the 340B program and how to access the medications they need.
- Collaboration with Pharmacies: Developing strong relationships with pharmacies to ensure efficient medication dispensing and minimize potential barriers to access.
- Data-Driven Decision Making: Utilizing data analytics to identify areas where 340B savings can be most effectively deployed to improve patient outcomes.
Pharmaceutical Company Perspectives
The 340B Drug Pricing Program, while intended to help safety-net hospitals and clinics serve vulnerable populations, presents a complex and often contentious issue for pharmaceutical manufacturers. Their perspectives are multifaceted, shaped by financial considerations, ethical obligations, and concerns about program integrity. Understanding these viewpoints is crucial to navigating the ongoing debate surrounding 340B.Pharmaceutical companies participating in the 340B program face a significant dilemma.
They are obligated to provide discounted drugs to covered entities, yet simultaneously need to maintain profitability and invest in research and development. This tension fuels much of the industry’s internal debate on the program’s effectiveness and fairness.
Arguments For and Against 340B from Pharmaceutical Companies
The pharmaceutical industry’s stance on 340B is not monolithic. Some companies support the program’s goals of expanding access to care for underserved communities. They may view their participation as a socially responsible contribution. However, many express concerns about the program’s potential for abuse and its impact on their bottom line. A key argument against 340B centers on the perceived lack of transparency and oversight, leading to concerns about inflated drug pricing and diversion of discounted medications.
The potential for large hospital systems to leverage the program for increased profit margins also fuels industry opposition.
Pharmaceutical Company Approaches to 340B Pricing and Distribution
Different pharmaceutical companies employ varying strategies in response to the 340B program. Some proactively engage with covered entities to ensure compliant practices and foster collaboration. Others might implement stricter controls over drug distribution to minimize the risk of diversion. Some companies may choose to limit their participation in the program or even withdraw entirely, depending on their assessment of the risks and benefits.
These diverse approaches reflect the range of perspectives within the industry and the absence of a standardized, universally accepted approach to managing 340B obligations.
Potential Impact of Policy Changes on Pharmaceutical Company Strategies
Policy changes regarding the 340B program could significantly alter pharmaceutical company strategies. For example, increased regulatory oversight and stricter enforcement of anti-diversion measures could lead companies to invest more in compliance programs and technology. Conversely, changes that weaken the program’s regulations might incentivize some companies to increase their participation, while others might maintain a cautious approach. Significant changes to reimbursement rates or the definition of covered entities could also necessitate significant adjustments in pricing models and distribution strategies, potentially leading to changes in the types of drugs offered at a discount or even a shift in focus to other market segments.
The inflated drug prices at 340B federal clinics are a major concern, especially considering the potential for discrepancies in pricing. Understanding these complexities is crucial, and a recent study, study widespread digital twins healthcare , highlights the power of data-driven solutions in healthcare. This kind of advanced analytics could potentially help us better monitor and regulate drug pricing within the 340B program, ensuring patients aren’t overcharged.
The potential for legal challenges to 340B regulations also necessitates proactive legal and strategic planning by pharmaceutical companies. For instance, a significant court ruling altering the scope of 340B could prompt a wave of adjustments in pricing and distribution strategies across the industry.
Ethical Considerations
Source: mdpi-res.com
The outrageous markups on pharmaceuticals for 340B federal clinics are a huge issue, impacting patient care and already strained budgets. It makes you wonder how these inflated costs compare to the struggles faced by healthcare workers, like those involved in the recent new york state nurse strike montefiore richmond university deals , where fair compensation was a major sticking point.
Ultimately, both situations highlight the deep flaws in our healthcare system’s financial structure, particularly regarding drug pricing and fair wages for essential medical professionals.
The 340B Drug Pricing Program, while designed to improve patient access to medications, presents several ethical dilemmas. Its inherent complexities, coupled with the significant financial incentives involved, create a landscape ripe for potential misuse and conflict. Transparency and accountability are crucial for ensuring the program’s integrity and fulfilling its intended purpose.The core ethical challenge lies in balancing the program’s goal of assisting safety-net hospitals with the potential for diverting funds away from direct patient care.
The substantial discounts offered under 340B can create a powerful incentive to maximize profits, potentially overshadowing the ethical obligation to prioritize patient needs. This tension needs careful consideration and robust oversight mechanisms.
Potential Ethical Dilemmas
The 340B program presents several potential ethical dilemmas. One key concern revolves around the potential for misuse of 340B savings. Funds intended to improve patient care could be diverted to other areas, potentially reducing the program’s overall effectiveness in reaching its intended beneficiaries. Another significant concern is the potential for inflated drug prices, as some hospitals may leverage the 340B discounts to increase their overall revenue, rather than directly reducing patient costs.
Furthermore, the lack of clear guidelines regarding the acceptable use of 340B savings can lead to varied interpretations and potentially unethical practices.
Transparency and Accountability in 340B
Transparency and accountability are essential for maintaining the ethical integrity of the 340B program. Open access to data on 340B purchases, drug pricing, and the allocation of savings is crucial for allowing independent scrutiny and identifying potential issues. Regular audits and robust enforcement mechanisms are needed to ensure compliance with program regulations and prevent misuse of funds. A publicly accessible database detailing 340B participation, drug purchases, and the resulting savings could greatly enhance transparency and accountability.
This would allow for independent verification of claims and facilitate identification of potential irregularities. For example, a comparison of 340B savings with actual improvements in patient care metrics could provide valuable insight into the program’s effectiveness and potential areas of improvement.
Conflicts of Interest within the 340B Program
The 340B program’s structure creates potential for conflicts of interest. Hospitals, motivated by financial incentives, may prioritize maximizing their 340B discounts over providing optimal patient care. Pharmaceutical companies, seeking to maintain profitability, may engage in practices that indirectly benefit hospitals participating in the program. For example, a pharmaceutical company might offer preferential pricing or other incentives to hospitals that purchase large quantities of their drugs, potentially creating an unfair advantage for those hospitals.
Furthermore, the lack of independent oversight could allow for undisclosed relationships between hospitals and pharmaceutical companies, creating further potential for conflicts of interest. This highlights the need for clear guidelines on acceptable interactions between hospitals and pharmaceutical companies, along with strict enforcement of those guidelines.
Best Practices for Ethical Conduct in Managing 340B Funds
Effective management of 340B funds requires a commitment to ethical conduct at all levels. This involves establishing clear policies and procedures, ensuring transparent financial reporting, and implementing robust internal controls.
- Develop a comprehensive written policy outlining the acceptable use of 340B savings, prioritizing patient care initiatives.
- Implement a system for tracking and monitoring 340B drug purchases and the allocation of resulting savings.
- Conduct regular internal audits to ensure compliance with program regulations and identify potential areas for improvement.
- Establish a transparent process for reporting 340B activities to relevant stakeholders, including patients, governing boards, and regulatory agencies.
- Provide regular training to staff on 340B regulations and ethical considerations related to the program.
- Foster a culture of ethical conduct and accountability throughout the organization.
Last Word: 340b Federal Clinics Pharma Drug Markups

Source: slideplayer.com
The 340B program, while intended to improve access to affordable medications for vulnerable populations, presents a tangled web of financial incentives, regulatory challenges, and ethical considerations. Understanding the complexities of drug markups within this program is crucial for ensuring its effectiveness and promoting equitable access to healthcare. Further discussion and potential regulatory adjustments are needed to strike a balance between supporting safety-net providers and maintaining fair pricing practices for all stakeholders.
FAQ Explained
What are the potential consequences of non-compliance with 340B regulations?
Non-compliance can lead to significant penalties, including fines, program termination, and even legal action. It can also damage a clinic’s reputation and jeopardize its ability to serve its patients.
How are 340B savings typically used by federal clinics?
Savings are often used to enhance patient care, expand services, improve infrastructure, and support staff training. Specific uses vary depending on the individual clinic’s needs and priorities.
Do all pharmaceutical companies participate equally in the 340B program?
No, pharmaceutical company participation and pricing strategies vary. Some companies are more supportive of the program than others, leading to inconsistencies in drug pricing across different manufacturers.
What role do patient advocacy groups play in the 340B debate?
Patient advocacy groups play a vital role in ensuring that the 340B program benefits patients. They advocate for transparency, accountability, and responsible use of 340B savings to maximize patient care.





