
What is Liquid Biopsy and How it Helps Detect Cancer?
What is liquid biopsy and how it helps in detecting cancer and monitoring treatment? It’s a game-changer in oncology, offering a less invasive way to diagnose and track cancer compared to traditional biopsies. Imagine a blood test that could reveal the presence of cancer cells, monitor their response to treatment, and even predict potential resistance – that’s the power of liquid biopsy.
This revolutionary technique analyzes bodily fluids, like blood or urine, for tiny fragments of tumor DNA, cancer cells, or other biomarkers to provide valuable insights into a patient’s cancer journey.
This post dives deep into the world of liquid biopsy, exploring its components, applications in cancer detection and treatment monitoring, limitations, and exciting future possibilities. We’ll look at real-world examples and address some common questions you might have about this groundbreaking technology.
Introduction to Liquid Biopsy
Imagine being able to detect cancer without the need for a traditional, invasive biopsy. That’s the promise of liquid biopsy, a groundbreaking approach that’s revolutionizing cancer diagnosis and treatment monitoring. It’s a less invasive, more convenient, and potentially more effective way to understand and manage the disease.Liquid biopsy is a minimally invasive procedure that analyzes bodily fluids, such as blood, urine, or cerebrospinal fluid, to detect cancer cells or their byproducts.
Liquid biopsy, a minimally invasive test, analyzes blood for cancer cells or DNA, revolutionizing early detection and treatment monitoring. It’s amazing how medical advancements are constantly improving, much like the decisions women are making about their reproductive health, as highlighted in this article about Karishma Mehta and egg freezing: karishma mehta gets her eggs frozen know risks associated with egg freezing.
Ultimately, both liquid biopsies and reproductive choices empower individuals to take control of their health journeys.
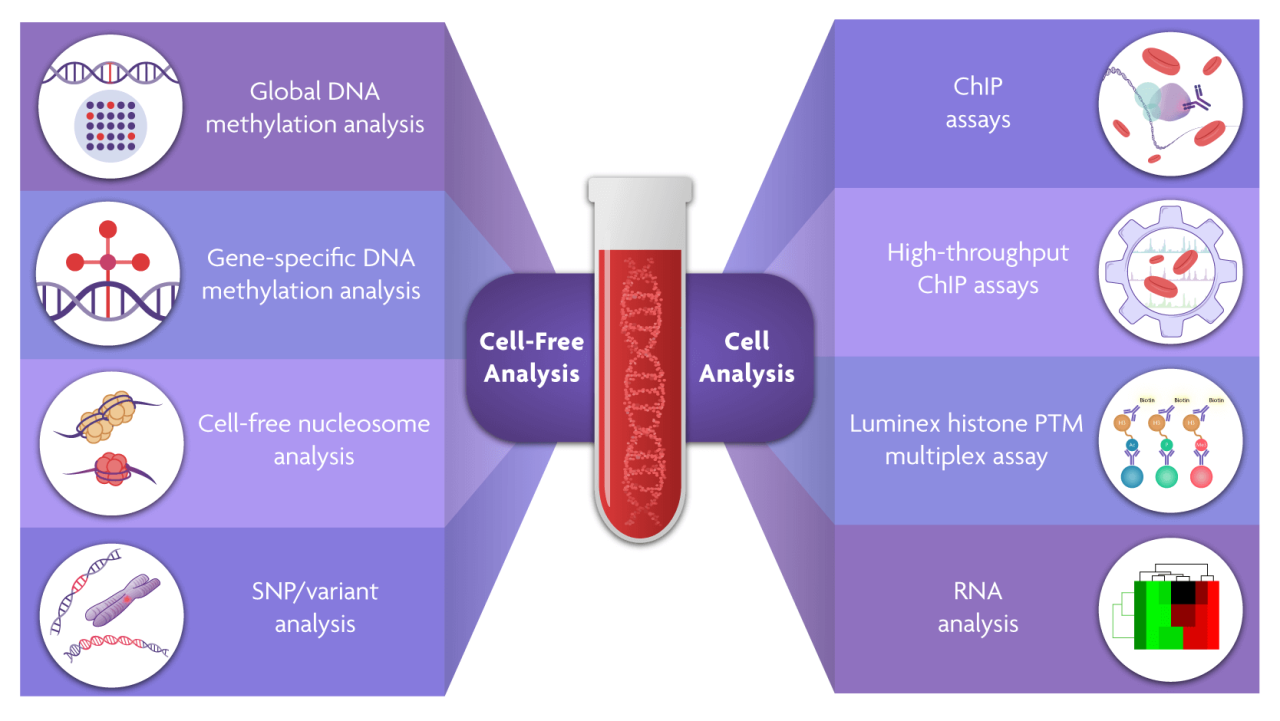
It’s essentially a “liquid” sample, unlike a traditional biopsy which involves removing a tissue sample. The core components of a liquid biopsy typically include circulating tumor cells (CTCs), circulating tumor DNA (ctDNA), exosomes, and other biomarkers found within these fluids. These components offer valuable insights into the cancer’s presence, characteristics, and response to treatment.
Advantages of Liquid Biopsy over Traditional Tissue Biopsies
Traditional tissue biopsies, while effective, involve invasive procedures with potential risks like bleeding, infection, and scarring. Liquid biopsies offer several key advantages. They are significantly less invasive, requiring only a simple blood draw, making them much more comfortable for patients. This minimally invasive nature also means they can be performed more frequently, allowing for closer monitoring of the cancer and its response to therapy.
Furthermore, liquid biopsies can provide a real-time snapshot of the tumor’s genetic makeup, offering a more comprehensive understanding of the disease’s heterogeneity – the fact that cancers often have diverse populations of cells with varying characteristics. This dynamic view contrasts with the static picture often provided by a single tissue biopsy, which may not represent the entire tumor.
This ability to repeatedly monitor ctDNA, for instance, is crucial in detecting minimal residual disease (MRD) after treatment, providing an early warning system for potential relapse. For example, a patient with colorectal cancer might undergo a liquid biopsy after surgery to detect any remaining cancerous cells circulating in their blood, allowing for prompt intervention if needed. This proactive approach helps improve treatment outcomes and overall patient survival.
Components of a Liquid Biopsy Sample
Liquid biopsy offers a minimally invasive way to analyze various components of blood to detect and monitor cancer. Understanding the different analytes present in a liquid biopsy sample is crucial for interpreting the results and making informed clinical decisions. These components provide a snapshot of the tumor’s characteristics and its response to treatment.
Circulating Tumor DNA (ctDNA)
ctDNA refers to fragments of DNA released by tumor cells into the bloodstream. These fragments carry the genetic mutations characteristic of the cancer, providing valuable information about the tumor’s genotype. Detection methods typically involve highly sensitive techniques like next-generation sequencing (NGS), which can identify specific mutations, copy number variations, and other genomic alterations. The sensitivity of ctDNA detection varies depending on the tumor type, tumor burden, and the sensitivity of the assay used.
For example, in advanced-stage lung cancer, ctDNA detection can be highly sensitive, while in early-stage cancers, detection rates may be lower.
Circulating Tumor Cells (CTCs)
CTCs are actual tumor cells that have shed from the primary tumor or metastases and are circulating in the bloodstream. These cells retain the characteristics of the original tumor, including their morphology, surface markers, and genetic profile. Detection methods involve enriching CTCs from the blood sample using various techniques, such as immunomagnetic separation or microfluidic devices. Once isolated, CTCs can be analyzed for genetic alterations, gene expression profiles, and drug sensitivity.
While CTC detection can be valuable for identifying and characterizing cancer cells, the low number of CTCs in the blood makes their detection less sensitive compared to ctDNA, especially in early-stage cancers. However, the ability to directly analyze intact tumor cells offers unique advantages.
Exosomes
Exosomes are small vesicles secreted by cells, including tumor cells. They contain a variety of molecules, such as proteins, lipids, and nucleic acids, reflecting the cellular state of origin. These molecules can act as biomarkers for cancer detection and monitoring. Detection methods involve isolating exosomes from the blood sample and analyzing their contents using techniques such as mass spectrometry for protein analysis or NGS for nucleic acid analysis.
Exosomes offer a promising area of liquid biopsy research, but standardization of detection methods and interpretation of results are still under development. Their sensitivity and specificity in cancer detection are still being investigated, and further research is needed to establish their clinical utility.
Comparison of Liquid Biopsy Components
The sensitivity and specificity of different liquid biopsy components vary significantly depending on several factors, including tumor type, stage, and the specific assay used. The following table provides a general comparison, keeping in mind that these are not absolute values and can vary considerably in different studies and clinical settings.
| Analyte | Sensitivity | Specificity | Advantages |
|---|---|---|---|
| ctDNA | High (especially in advanced cancers) | Generally high | Non-invasive, reflects tumor genomic heterogeneity |
| CTCs | Moderate to low | High | Provides information on tumor cell biology and heterogeneity |
| Exosomes | Low to moderate (currently under investigation) | Under investigation | Potential for early detection and monitoring of treatment response |
Liquid Biopsy in Cancer Detection
Liquid biopsy is revolutionizing cancer detection and treatment monitoring by offering a less invasive and more accessible alternative to traditional tissue biopsies. It allows for the analysis of circulating tumor cells (CTCs), circulating tumor DNA (ctDNA), and other biomarkers present in bodily fluids like blood, urine, or cerebrospinal fluid, providing valuable insights into the presence, type, and progression of cancer.
This minimally invasive approach offers significant advantages, especially in early detection and ongoing monitoring of treatment response.
Early Cancer Detection with Liquid Biopsy
Early detection is crucial for successful cancer treatment. Liquid biopsy offers a significant advantage in this regard because it can detect minute amounts of cancer-related material in the bloodstream, often before tumors are large enough to be detected by conventional imaging techniques like CT scans or MRIs. This early detection capability significantly improves the chances of successful treatment and increases patient survival rates.
The sensitivity of liquid biopsy tests is constantly improving, leading to earlier and more accurate diagnoses. For instance, studies have shown the potential for detecting early-stage lung cancer using ctDNA analysis with higher accuracy than traditional screening methods in high-risk individuals.
Identifying Different Cancer Types Using Liquid Biopsy
Liquid biopsy isn’t limited to detecting the mere presence of cancer; it can also help identify the specific type of cancer. Different cancers shed unique genetic and molecular signatures into the bloodstream. By analyzing these signatures—through techniques like next-generation sequencing (NGS)—liquid biopsy can distinguish between various cancer types, guiding clinicians towards more targeted diagnostic and treatment strategies. For example, the presence of specific gene mutations or epigenetic alterations in ctDNA can help differentiate between different subtypes of lung cancer (e.g., adenocarcinoma, squamous cell carcinoma), leading to more personalized treatment plans.
Examples of Cancers Where Liquid Biopsy Shows Promise
Liquid biopsy has shown exceptional promise across a range of cancers. In lung cancer, it’s proving invaluable for early detection, monitoring treatment response, and identifying resistance mutations. Similarly, in colorectal cancer, liquid biopsy aids in detecting recurrence and guiding treatment decisions. For metastatic cancers, where obtaining tissue samples is challenging, liquid biopsy offers a less invasive alternative for monitoring disease progression and tailoring therapy.
In breast cancer, ctDNA analysis can help predict the risk of recurrence and guide adjuvant therapy decisions. The application of liquid biopsy is rapidly expanding to encompass various other cancer types, including prostate, pancreatic, and ovarian cancers.
Flowchart Illustrating Liquid Biopsy for Cancer Screening
The process of using liquid biopsy for cancer screening can be visualized as follows:[A flowchart would be inserted here. It would begin with a box labeled “Patient presents for cancer screening,” leading to a box labeled “Blood sample collected.” This would then branch to a box labeled “Sample processing and DNA extraction,” which would connect to a box labeled “ctDNA analysis (NGS, etc.).” The results from the ctDNA analysis would then lead to two possible outcomes: a box labeled “Cancer detected; further investigation needed,” and a box labeled “No cancer detected; follow-up recommended.”] The flowchart visually represents the sequential steps involved in liquid biopsy for cancer screening, from sample collection to result interpretation.
The specific tests used within the ctDNA analysis box would depend on the suspected cancer type and the clinical context.
Monitoring Cancer Treatment with Liquid Biopsy

Source: rsc.org
Liquid biopsy offers a powerful new tool for monitoring how cancer patients respond to treatment. Unlike traditional methods like tissue biopsies, which are invasive and require surgery, liquid biopsy uses blood samples to track cancer-related molecules, such as circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and exosomes. This non-invasive approach allows for more frequent monitoring, providing valuable insights into treatment efficacy and the emergence of resistance.Liquid biopsy helps monitor treatment response by detecting changes in the levels of ctDNA, CTCs, and other biomarkers.
A decrease in ctDNA levels, for example, might indicate that the treatment is effectively shrinking the tumor. Conversely, an increase in ctDNA levels or the appearance of new mutations could signal that the cancer is progressing or developing resistance to the therapy. This real-time feedback allows oncologists to adjust treatment strategies promptly, potentially improving patient outcomes.
Detection of Treatment Resistance Using Liquid Biopsy
The ability of liquid biopsy to detect treatment resistance is a significant advantage. Traditional methods often rely on imaging techniques like CT scans or MRI, which may not detect subtle changes indicative of resistance until the disease has progressed considerably. Liquid biopsy, however, can detect the emergence of drug-resistance mutations in ctDNA much earlier. For instance, in patients receiving EGFR-targeted therapy for lung cancer, the appearance of T790M mutation in ctDNA can signal the development of resistance, prompting a switch to a different therapy.
Similarly, in melanoma patients treated with BRAF inhibitors, the detection of BRAF V600E mutations can indicate resistance. Early detection of these mutations allows for timely intervention, preventing disease progression and improving survival rates.
Guiding Treatment Decisions and Personalizing Therapy
Liquid biopsy plays a crucial role in guiding treatment decisions and personalizing cancer therapy. By analyzing the genetic makeup of the tumor through ctDNA, oncologists can identify specific mutations driving cancer growth. This information can be used to select the most appropriate targeted therapy, maximizing the chances of success while minimizing side effects. For example, patients with a specific mutation in the HER2 gene in breast cancer might benefit from targeted therapy with Herceptin, whereas patients without this mutation might respond better to a different treatment approach.
Liquid biopsy also allows for continuous monitoring of the tumor’s response, enabling adjustments to therapy based on the patient’s individual needs. This personalized approach can significantly improve outcomes and reduce the burden of ineffective treatments.
Comparison of Liquid Biopsy’s Role in Monitoring Various Cancer Treatments
The table below summarizes the role of liquid biopsy in monitoring different cancer treatments.
| Treatment Type | How Liquid Biopsy Monitors Response | Examples of Resistance Detection | Treatment Adjustments Guided by Liquid Biopsy |
|---|---|---|---|
| Chemotherapy | Monitoring ctDNA levels to assess tumor burden reduction. | Increase in ctDNA despite chemotherapy, emergence of new mutations conferring resistance. | Changing chemotherapy regimen, adding targeted therapy. |
| Immunotherapy | Monitoring immune cell populations (e.g., T cells) and inflammatory markers. Tracking ctDNA levels for tumor response. | Lack of decrease in ctDNA, immune escape mechanisms detected through ctDNA analysis. | Combination therapy with chemotherapy or targeted therapy, exploring alternative immunotherapy approaches. |
| Targeted Therapy | Monitoring for specific mutations associated with resistance. Tracking ctDNA levels. | Emergence of resistance mutations (e.g., T790M in EGFR-mutated lung cancer). | Switching to a different targeted therapy, adding chemotherapy or immunotherapy. |
Limitations and Future Directions of Liquid Biopsy
Liquid biopsy, while a revolutionary tool in cancer diagnostics and monitoring, is not without its limitations. Current technology presents several hurdles that need to be overcome to fully realize its potential. Understanding these limitations is crucial for responsible implementation and for guiding future research directions.Current limitations of liquid biopsy technology primarily revolve around sensitivity, specificity, and standardization. The concentration of circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and exosomes in blood samples can be incredibly low, leading to false negative results.
Furthermore, distinguishing between cancer-related biomarkers and those originating from normal cells or other conditions remains a challenge, leading to false positives. These issues significantly impact the reliability and clinical utility of liquid biopsy in various cancer types and stages.
Sensitivity and Specificity Challenges
The sensitivity of liquid biopsy, meaning its ability to detect cancer when it is present, is affected by several factors. Tumor heterogeneity, the variation in genetic makeup within a single tumor, can lead to missed detection if the sampled ctDNA doesn’t represent the entire tumor burden. Similarly, the low concentration of CTCs and exosomes in the bloodstream makes their detection difficult, especially in early-stage cancers.
Improving the sensitivity of liquid biopsy assays is a critical area of ongoing research, focusing on more sensitive detection methods and pre-analytical handling procedures that minimize ctDNA degradation. The specificity, the ability to correctly identify cancer and rule out other conditions, is equally important. Non-cancerous conditions can also release similar biomarkers into the bloodstream, leading to false positive results.
Liquid biopsy is a game-changer in cancer care, offering a less invasive way to detect cancer cells and monitor treatment response through blood samples. Understanding how effective treatments are is crucial, much like understanding the risk factors that make stroke more dangerous is vital for prevention. Early detection and monitoring with liquid biopsy can significantly improve patient outcomes, giving us a powerful tool against this devastating disease.
Developing more sophisticated algorithms and using multiple biomarkers in combination can enhance the specificity of liquid biopsy tests. For example, combining ctDNA analysis with CTC enumeration might improve diagnostic accuracy compared to using either method alone.
Standardization and Inter-laboratory Variability
Standardization is a major obstacle hindering the widespread adoption of liquid biopsy. Different laboratories may use varying sample processing techniques, assay platforms, and analytical methods, leading to inconsistencies in results. This inter-laboratory variability makes it difficult to compare data across studies and to establish universally accepted clinical guidelines for the use of liquid biopsy. Efforts are underway to develop standardized protocols for sample collection, processing, and analysis to improve the reproducibility and reliability of liquid biopsy tests.
This includes the development of standardized reference materials and quality control measures to ensure consistent performance across different laboratories. The creation of large, well-annotated datasets, including both positive and negative controls, will also be crucial in establishing standardized interpretation guidelines.
Future Directions and Technological Advancements
Despite the limitations, the future of liquid biopsy is bright. Advancements in technology are continuously improving the sensitivity, specificity, and cost-effectiveness of liquid biopsy assays. Next-generation sequencing (NGS) technologies are enabling the detection of increasingly smaller amounts of ctDNA, allowing for earlier and more accurate cancer detection. Furthermore, the development of more sophisticated algorithms and machine learning techniques is enhancing the ability to analyze complex datasets and identify subtle patterns indicative of cancer.
Research is also focusing on the development of new biomarkers that are more specific to cancer and less susceptible to interference from other sources. This includes exploring novel circulating biomarkers, such as circulating microRNAs and proteins, which may provide additional information about cancer progression and response to therapy.
Beyond Cancer Detection and Monitoring
The potential applications of liquid biopsy extend far beyond cancer detection and monitoring. It shows promise in other areas of medicine, including the diagnosis and monitoring of infectious diseases, autoimmune disorders, and cardiovascular diseases. For example, liquid biopsy techniques are being explored for the early detection of infectious agents in blood samples, which could lead to faster and more effective treatment.
Similarly, the detection of specific biomarkers in blood samples may aid in the diagnosis and management of autoimmune diseases. In cardiovascular disease, liquid biopsy could help identify individuals at risk of developing heart attacks or strokes by detecting circulating biomarkers indicative of plaque rupture or inflammation. The versatility of liquid biopsy makes it a valuable tool with the potential to revolutionize many areas of healthcare beyond oncology.
The development of more sensitive and specific assays, coupled with improved data analysis techniques, will unlock the full potential of liquid biopsy across various medical fields.
Liquid biopsies are amazing; they analyze blood samples to detect cancer DNA, offering earlier diagnosis and treatment monitoring. It’s fascinating how such minimally invasive techniques are revolutionizing oncology, a stark contrast to the complexities involved in managing conditions like Tourette Syndrome in children, for which helpful strategies can be found here: strategies to manage Tourette syndrome in children.
Returning to liquid biopsies, their ability to track treatment response makes them invaluable for personalized cancer care.
Illustrative Example
Source: com.cn
This case study illustrates how liquid biopsy helped guide the diagnosis and treatment of a patient with metastatic lung cancer. The example uses hypothetical data for illustrative purposes but reflects the real-world applications of this technology.
Sarah, a 58-year-old woman, presented with persistent coughing and shortness of breath. Initial imaging revealed a suspicious lung mass, prompting a biopsy. The tissue biopsy confirmed the presence of non-small cell lung cancer (NSCLC), specifically adenocarcinoma. However, staging revealed the cancer had already metastasized to the lymph nodes and liver.
Initial Diagnosis and Treatment Decisions Based on Liquid Biopsy, What is liquid biopsy and how it helps in detecting cancer and monitoring treatment
Alongside the traditional tissue biopsy, Sarah underwent a liquid biopsy. This involved a simple blood draw, which was analyzed for circulating tumor DNA (ctDNA) and circulating tumor cells (CTCs). The ctDNA analysis revealed a specific EGFR mutation (exon 19 deletion). This genetic information was crucial because it indicated that Sarah would likely respond well to targeted therapy with EGFR tyrosine kinase inhibitors (TKIs), such as gefitinib or erlotinib.
The absence of significant CTCs in the initial liquid biopsy suggested a relatively low tumor burden at the time of diagnosis.
Monitoring Treatment Response with Liquid Biopsy
Sarah began treatment with gefitinib. Regular liquid biopsies were performed every three months to monitor her response. The initial post-treatment liquid biopsy showed a significant decrease in ctDNA levels, indicating a good response to the therapy. This confirmed the effectiveness of the targeted therapy and avoided the need for more invasive and frequent imaging scans. After six months, the ctDNA levels remained low, and Sarah experienced a significant improvement in her symptoms.
Detection of Treatment Resistance Using Liquid Biopsy
However, after twelve months of treatment, Sarah’s ctDNA levels began to rise again, and she started experiencing a recurrence of her symptoms. A subsequent liquid biopsy revealed the emergence of a secondary T790M mutation in the EGFR gene, which conferred resistance to gefitinib. This information was critical, as it allowed for a swift change in treatment strategy. Sarah was switched to a third-generation EGFR TKI, osimertinib, which is effective against the T790M mutation.
Impact on Patient Outcome
The continuous monitoring provided by liquid biopsy allowed for early detection of treatment resistance and a timely adjustment to Sarah’s therapy. This proactive approach significantly prolonged her progression-free survival and improved her quality of life. While the cancer is still present, Sarah’s condition is currently well-managed thanks to the insights gained from repeated liquid biopsies. The minimally invasive nature of the procedure also minimized the burden of repeated biopsies and extensive imaging.
Closing Notes: What Is Liquid Biopsy And How It Helps In Detecting Cancer And Monitoring Treatment
Liquid biopsy is rapidly transforming cancer care, offering a less invasive, more efficient, and potentially more effective approach to diagnosis and treatment monitoring. While challenges remain in standardization and widespread accessibility, the future of liquid biopsy looks incredibly bright. From earlier detection and personalized therapies to predicting treatment response and identifying resistance, liquid biopsy holds immense promise for improving cancer outcomes and offering patients a better chance at a healthier future.
It’s a field constantly evolving, and we can expect even more groundbreaking advancements in the years to come.
Questions Often Asked
What are the risks associated with a liquid biopsy?
The risks are generally minimal, similar to a standard blood test. There’s a small chance of bruising or discomfort at the puncture site. The procedure itself is non-invasive.
Is liquid biopsy suitable for all types of cancer?
While liquid biopsy is showing promise for many cancer types, its effectiveness varies. Research is ongoing to determine its optimal application across different cancers and stages.
How much does a liquid biopsy cost?
The cost can vary depending on the specific tests performed and the laboratory conducting the analysis. It’s generally more expensive than a standard blood test, but costs are likely to decrease as the technology becomes more widely adopted.
How long does it take to get the results of a liquid biopsy?
Turnaround times vary depending on the laboratory and the complexity of the tests. Results typically take several days to a few weeks.
