Appellate Court Rules on 340B, Novartis, and Specialty Pharmacies
Appellate court rules drugmakers 340b novartis pharmacies specialty – Appellate Court Rules on 340B, Novartis, and Specialty Pharmacies – this headline alone throws a legal bombshell into the already complex world of pharmaceutical pricing! The recent court decision regarding the 340B Drug Pricing Program, Novartis, and specialty pharmacies has sent ripples through the healthcare industry. This ruling has major implications for hospitals, drug manufacturers, and, most importantly, patients.
Let’s dive into the details and unpack what this all means.
The 340B program itself aims to help safety-net hospitals and clinics provide affordable care to underserved communities. But the program’s intricacies, coupled with the significant financial stakes involved, have led to ongoing disputes. Novartis, a major pharmaceutical player, found itself at the center of a legal battle highlighting the tensions between drug manufacturers and participating entities. The role of specialty pharmacies, which often handle expensive medications, further complicates the issue.
This court ruling attempts to clarify these complexities, but the long-term consequences remain to be seen.
The 340B Program and Drug Manufacturers

Source: ytimg.com
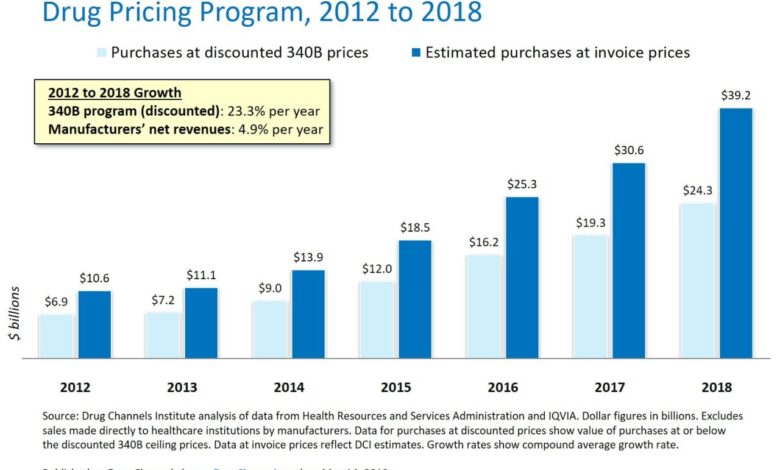
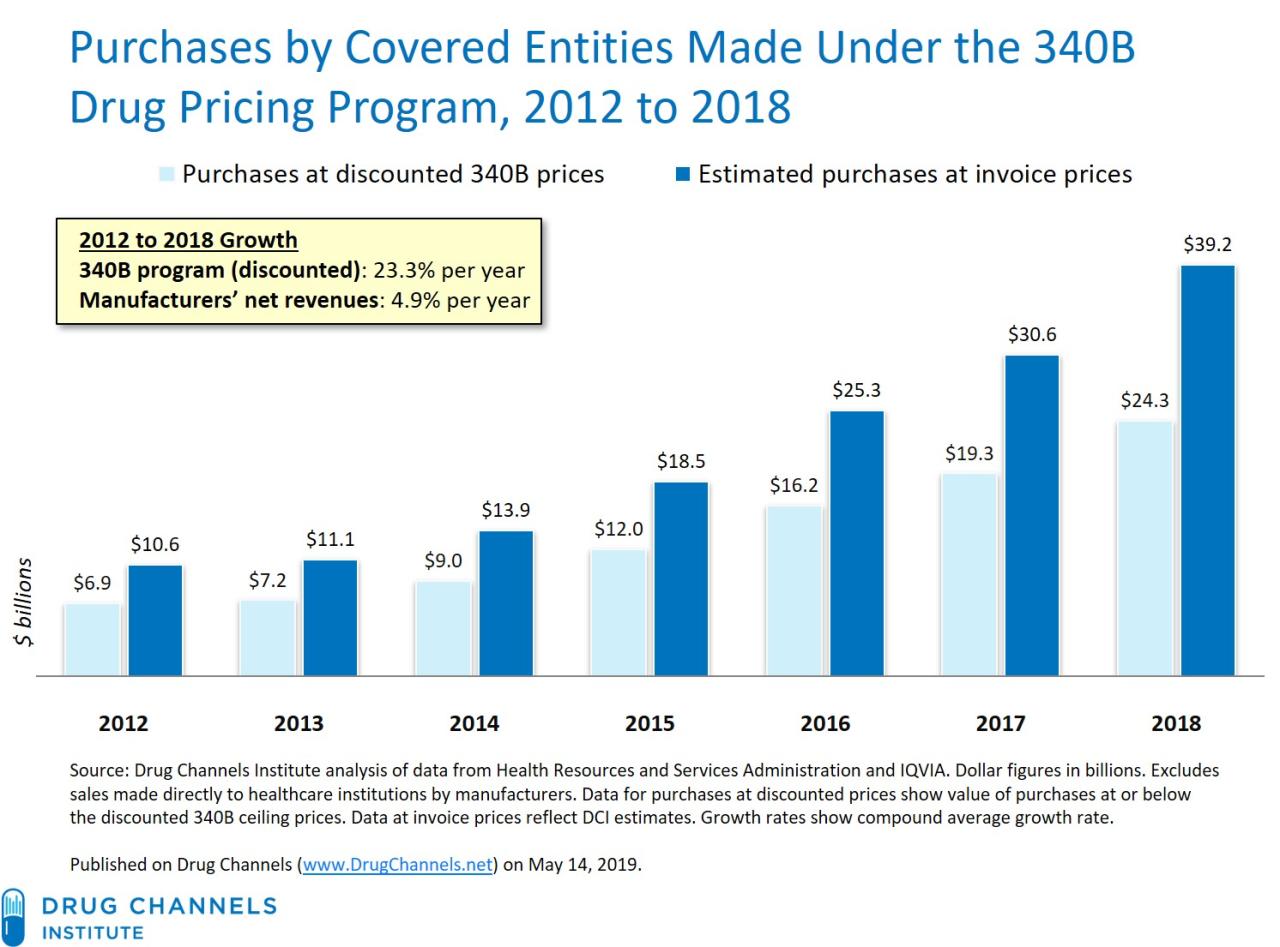
The 340B Drug Pricing Program is a complex and often controversial aspect of the US healthcare system. It aims to stretch limited healthcare resources by providing discounted prescription drugs to eligible healthcare organizations serving vulnerable populations. This program significantly impacts both drug manufacturers, who provide the discounted medications, and the hospitals and clinics that receive them. Understanding its intricacies is crucial for navigating the landscape of pharmaceutical pricing and healthcare access.
The 340B Drug Pricing Program: Purpose and Operation, Appellate court rules drugmakers 340b novartis pharmacies specialty
The 340B program, established under the Public Health Service Act, requires drug manufacturers to provide outpatient drugs to covered entities at significantly reduced prices. The intended purpose is to allow these entities, which often serve disproportionately high numbers of uninsured or low-income patients, to stretch their budgets and provide more comprehensive care. This means that hospitals and clinics can use the savings generated from 340B discounts to expand services, invest in infrastructure, and improve patient care, ultimately benefiting vulnerable communities.
The program’s effectiveness, however, is a subject of ongoing debate and scrutiny.
Participation Requirements for Drug Manufacturers and Covered Entities
Drug manufacturers participating in the 340B program must contract with the Health Resources and Services Administration (HRSA). This contract obligates them to provide discounts on a list of covered outpatient drugs to eligible entities. Failure to comply can result in significant penalties. Covered entities, on the other hand, must meet specific criteria to qualify for the program. These criteria typically include serving a significant percentage of low-income patients and demonstrating a commitment to providing comprehensive healthcare services in underserved areas.
The recent appellate court ruling on drugmakers and 340B Novartis pharmacies is a significant development in healthcare access, especially considering the complexities of specialty drug pricing. It’s fascinating to contrast this with breakthroughs like the FDA’s approval of clinical trials for pig kidney transplants in humans, as reported by this article. This highlights the ongoing push for innovation, even as we grapple with the existing challenges of equitable drug distribution via programs like 340B.
The HRSA maintains a list of approved covered entities and regularly audits them to ensure continued compliance with program requirements. This rigorous process aims to prevent fraud and abuse within the program.
Financial Implications of 340B Participation
For drug manufacturers, participation in the 340B program means a reduction in revenue from sales to covered entities. The extent of this reduction varies depending on the specific drugs and the volume of sales to 340B entities. However, manufacturers generally receive higher reimbursement rates from commercial insurers and government programs for the same drugs. Some manufacturers argue that the net impact on their overall profitability is minimal, while others express concerns about the financial burden of the program.
For covered entities, the 340B discounts provide substantial financial relief, allowing them to allocate resources to patient care and community outreach programs. However, the program’s complex administrative requirements and the potential for audits can also present financial challenges.
Pricing Comparison: 340B vs. Non-340B Entities
The following table compares the pricing structures for 340B covered entities and non-covered entities. It’s important to note that actual prices can vary significantly depending on the specific drug, manufacturer, and payer. This is a simplified illustration for comparative purposes only.
| Drug | Manufacturer’s Wholesale Acquisition Cost (WAC) | 340B Price (Estimate) | Non-340B Price (Estimate) |
|---|---|---|---|
| Example Drug A | $100 | $60 | $100 |
| Example Drug B | $200 | $120 | $200 |
| Example Drug C | $50 | $30 | $50 |
Novartis’ Involvement in the 340B Program
Novartis, like other major pharmaceutical companies, participates in the 340B Drug Pricing Program, a complex system designed to stretch healthcare resources for safety-net providers. Their involvement has been marked by both cooperation and periods of tension, reflecting the ongoing debate surrounding the program’s implementation and interpretation. Understanding Novartis’s role requires examining their historical participation, public statements, and any legal challenges they’ve faced.Novartis’s participation in the 340B program involves supplying medications at discounted prices to eligible healthcare providers.
This participation is governed by contracts and agreements with individual 340B covered entities, the specifics of which are generally not publicly available due to confidentiality clauses. However, the general principle is that Novartis commits to providing certain medications at the 340B ceiling price, as mandated by the program’s regulations. This ensures that safety-net hospitals and clinics can offer vital medications to vulnerable populations at lower costs.
Novartis’ 340B Contracts and Agreements
While the exact details of Novartis’ contracts with 340B covered entities are confidential, we can infer the general structure. These agreements likely specify the drugs covered under the 340B discount, the pricing structure, reporting requirements, and mechanisms for dispute resolution. They probably also include clauses related to compliance with program rules and regulations, including those regarding contract pharmacies and proper dispensing practices.
The lack of public access to these documents limits detailed analysis, but the general framework aligns with industry standards for participation in the 340B program.
Novartis and 340B: Controversies and Legal Challenges
Novartis, like other pharmaceutical manufacturers, has faced challenges related to the 340B program. These challenges often center on disagreements over eligibility criteria for covered entities, the proper calculation of discounts, and the appropriate handling of contract pharmacies. While specific lawsuits involving Novartis are not readily available in public domain databases, the industry as a whole has seen numerous legal battles over interpretations of 340B regulations.
These disputes highlight the ongoing complexities of balancing the program’s goals with the financial interests of drug manufacturers. One common area of contention is the definition of a “covered entity” and the appropriate use of contract pharmacies to dispense 340B drugs.
Evolution of Novartis’ 340B Policies
Novartis’ policies regarding the 340B program have likely evolved over time in response to changing regulations, industry practices, and legal challenges. While specific policy changes are not readily available publicly, it’s reasonable to assume that Novartis has adjusted its internal processes and contracts to reflect updates to the 340B program’s rules and guidance. This evolution likely includes refinements in contract language, improved tracking of 340B sales, and strengthened compliance procedures to mitigate potential risks associated with non-compliance.
The company’s public statements and participation in industry discussions on the 340B program can offer some insights into their evolving approach.
Specialty Pharmacies and the 340B Program
Specialty pharmacies play a crucial, albeit complex, role in the 340B Drug Pricing Program. Their unique focus on high-cost, specialized medications creates both significant opportunities and considerable challenges within the program’s framework. Understanding these nuances is vital for ensuring program integrity and patient access to life-saving therapies.
Unique Challenges Faced by Specialty Pharmacies Participating in the 340B Program
Specialty pharmacies face a unique set of challenges within the 340B program, primarily stemming from the complexities of handling expensive, high-risk medications. These challenges include navigating intricate reimbursement processes with payers, managing stringent medication handling and storage requirements, and ensuring patient adherence to complex treatment regimens. The high cost of medications necessitates robust inventory management systems to minimize waste and maximize efficiency.
Additionally, the specialized nature of the drugs often requires highly trained staff capable of addressing patient inquiries and managing potential adverse effects. The regulatory landscape is also complex, requiring meticulous record-keeping and compliance with multiple federal and state regulations.
Comparison of Dispensing Practices Between Specialty and Retail Pharmacies within the 340B Context
While both specialty and retail pharmacies participate in the 340B program, their dispensing practices differ significantly. Retail pharmacies primarily dispense common medications for chronic conditions and acute illnesses, often with relatively straightforward dispensing protocols. Specialty pharmacies, however, handle medications requiring specialized handling, storage, and administration. This includes medications requiring refrigeration, protection from light, or specific preparation before administration. The dispensing process itself often involves more extensive patient counseling, medication reconciliation, and monitoring of therapeutic outcomes.
Furthermore, specialty pharmacies frequently engage in prior authorization processes with insurance companies, a task less common in retail settings. The complexity of these processes increases the administrative burden on specialty pharmacies participating in 340B.
The Role of Specialty Pharmacies in Providing Access to Expensive Medications Under the 340B Program
Specialty pharmacies play a vital role in expanding access to expensive medications for vulnerable populations through the 340B program. Many of the drugs dispensed by specialty pharmacies are prohibitively expensive for uninsured or underinsured patients. The 340B discounts allow covered entities to acquire these medications at significantly reduced prices, making them affordable for their patients. This is particularly crucial for medications used to treat complex conditions like cancer, multiple sclerosis, and rheumatoid arthritis, where timely access to therapy is paramount.
Without the 340B program and the participation of specialty pharmacies, many patients would be unable to afford these life-saving treatments.
Hypothetical Scenario Illustrating Potential for Fraud or Abuse within Specialty Pharmacies Utilizing the 340B Program
Consider a hypothetical scenario where a specialty pharmacy participating in the 340B program intentionally misclassifies patients to increase the number of 340B-eligible prescriptions. The pharmacy might falsely claim that patients meet the criteria for 340B coverage, even if they don’t. This could involve manipulating patient addresses or billing information to make them appear eligible. Another potential abuse could involve upcoding, where the pharmacy bills for a more expensive medication than what was actually dispensed, thereby increasing their profits.
The appellate court’s ruling on drugmakers, 340B, Novartis, and specialty pharmacies is a huge deal, impacting healthcare costs significantly. This comes at a time when Robert F. Kennedy Jr. cleared a key hurdle in his bid to become HHS Secretary, as reported here. His potential appointment could drastically shift the landscape of pharmaceutical regulation, potentially influencing future decisions related to the 340B program and its implications for drug pricing.
Such fraudulent activities could involve collusion with healthcare providers to generate false prescriptions or documentation. These actions not only violate the 340B program’s rules but also defraud government programs and potentially harm patients by denying them access to appropriate care. This illustrates the importance of robust oversight and auditing mechanisms to ensure the integrity of the program.
Appellate Court Ruling and its Implications
The recent appellate court decision regarding Novartis and the 340B drug pricing program has sent shockwaves through the pharmaceutical industry and healthcare system. The ruling clarifies, to some extent, the complex relationship between drug manufacturers, 340B covered entities, and specialty pharmacies, but also leaves many questions unanswered and opens the door to further legal challenges. The core issue revolves around the legality of Novartis’s actions in restricting the flow of discounted 340B drugs to specialty pharmacies serving 340B covered entities.
Summary of the Appellate Court Ruling
The appellate court largely upheld the lower court’s decision, finding that Novartis’s actions to limit the 340B drug discounts to specialty pharmacies were, at least in part, unlawful. The court’s reasoning centered on the interpretation of the 340B program’s regulations and the contractual agreements between Novartis and the participating pharmacies. The specific details of the ruling will vary depending on the specific case being referenced, but generally, the court found that Novartis could not unilaterally restrict the participation of specialty pharmacies in the 340B program, as long as those pharmacies were legitimately contracted with 340B covered entities and adhered to the program’s regulations.
The ruling did not entirely invalidate Novartis’s contract stipulations, but it significantly narrowed their scope and impact.
Arguments Presented by Novartis and the Plaintiffs
Novartis argued that they had the right to control the distribution of their discounted drugs under the 340B program to prevent fraud and abuse. They presented evidence suggesting that some specialty pharmacies were engaging in practices that violated the spirit, if not the letter, of the 340B program, such as diverting drugs to non-eligible patients or improperly billing for services.
The plaintiffs, representing 340B covered entities and specialty pharmacies, countered that Novartis’s actions constituted an unlawful attempt to circumvent the program’s intent to provide affordable medications to vulnerable populations. They argued that Novartis’s restrictions unfairly limited patient access to essential medications and that the company’s concerns about fraud could be addressed through more appropriate means, such as increased oversight and collaboration with the Health Resources and Services Administration (HRSA), the agency that oversees the 340B program.
Legal Reasoning of the Court
The court’s decision rested heavily on its interpretation of the statutory language governing the 340B program and the contractual agreements between Novartis and the pharmacies. The judges carefully examined the language used in both the statute and the contracts to determine whether Novartis’s actions were consistent with the program’s overall goals and legal framework. The court likely emphasized that the 340B program is intended to benefit patients and healthcare providers, and that actions restricting access to discounted medications should be carefully scrutinized.
The court’s reasoning likely included a detailed analysis of the relevant case law and regulatory guidance, demonstrating a thorough understanding of the complexities surrounding the 340B program. A crucial aspect of the legal reasoning may have been a consideration of whether Novartis’s actions were unduly burdensome or restrictive on access to care for vulnerable populations.
Comparison with Previous Legal Precedents
This ruling builds upon, and in some ways departs from, previous legal precedents concerning the 340B program. Prior cases have addressed various aspects of the program’s complexities, including disputes over eligibility, drug pricing, and contract interpretation. This specific case, however, is notable for its focus on the role of specialty pharmacies and the extent to which manufacturers can control their distribution networks within the 340B context.
While some previous rulings may have favored manufacturers’ control over their products, this case seems to lean towards a stronger emphasis on ensuring patient access to affordable medications as a core tenet of the 340B program. The decision may lead to increased scrutiny of manufacturer contracts and practices in the future, potentially influencing future litigation.
The recent appellate court ruling on drugmakers, 340B, Novartis, and specialty pharmacies is a complex issue, highlighting the challenges in healthcare pricing. It got me thinking about how seemingly unrelated areas of medicine intersect; for instance, I read an interesting article about whether an eye test can detect dementia risk in older adults – you can check it out here: can eye test detect dementia risk in older adults.
The implications of early detection, much like the implications of fair drug pricing, are huge for patient care and resource allocation. Ultimately, both situations emphasize the importance of innovative solutions within the healthcare system.
Impact on Healthcare Providers and Patients

Source: chroniccarealliance.org
The appellate court’s ruling on 340B drug pricing has significant ramifications for healthcare providers and the patients they serve. The decision, which limits the discounts available to covered entities under the 340B program, will likely impact the affordability and accessibility of medications, particularly for vulnerable populations who rely on these hospitals and clinics. The financial implications for healthcare providers are also substantial, potentially forcing difficult choices regarding resource allocation and patient care.The ruling’s effect on medication affordability and accessibility is multifaceted.
Hospitals and clinics participating in the 340B program often rely on the discounts to subsidize care for low-income and uninsured patients. Reduced discounts mean higher drug costs for these facilities, potentially leading to increased patient out-of-pocket expenses or a reduction in the availability of certain medications. This could disproportionately affect patients with chronic conditions requiring expensive medications, pushing them towards less effective or more costly treatment options.
The reduced availability of affordable medications could also lead to poorer health outcomes and increased hospital readmissions, creating a ripple effect throughout the healthcare system.
Financial Stability of Hospitals and Covered Entities
The financial impact on hospitals and other covered entities is potentially severe. Many hospitals, particularly those serving a large proportion of low-income patients, rely on 340B savings to offset operating losses. The reduced drug discounts could significantly strain their budgets, forcing them to make difficult choices, such as reducing staff, cutting services, or increasing charges for other services.
This could particularly impact rural hospitals and safety-net hospitals, which often have thinner margins and serve vulnerable populations. For example, a rural hospital heavily reliant on 340B discounts might face budget shortfalls that force them to limit access to specialized care or reduce staffing levels, potentially impacting the quality of care provided. This scenario is not hypothetical; numerous rural hospitals across the country are already struggling financially, and this ruling could push some over the edge.
Strategies for Healthcare Providers to Adapt
Healthcare providers will need to adopt various strategies to mitigate the financial impact of the ruling. This could involve exploring alternative cost-saving measures, such as negotiating better contracts with other suppliers, improving efficiency in medication dispensing, and focusing on preventative care to reduce the need for expensive treatments. Diversifying revenue streams through expanded services or increased focus on outpatient care could also help.
Additionally, advocating for legislative changes to support the 340B program or seeking additional funding from state or federal governments could provide some relief. Strengthening relationships with pharmaceutical companies to negotiate better pricing outside of the 340B program is another potential avenue for cost reduction. Finally, improved inventory management and waste reduction can also contribute to better financial outcomes.
Potential Long-Term Consequences for the Healthcare System
The long-term consequences of this ruling are uncertain but potentially significant.
- Reduced access to affordable medications for vulnerable populations.
- Increased financial strain on hospitals and clinics, potentially leading to closures or service reductions.
- Deterioration in the quality of care provided to low-income and uninsured patients.
- Increased healthcare costs for patients and the overall healthcare system.
- Potential exacerbation of health disparities between different socioeconomic groups.
Future of the 340B Program: Appellate Court Rules Drugmakers 340b Novartis Pharmacies Specialty
Source: blogspot.com
The recent appellate court ruling regarding Novartis and 340B pharmacies has sent ripples through the healthcare system, leaving the future of the program uncertain. This decision, and potential future legal challenges, will undoubtedly spur legislative and regulatory responses, prompting a critical reassessment of the program’s design, effectiveness, and overall fairness. The coming years will be crucial in shaping the 340B program’s trajectory, impacting both the providers who rely on it and the patients they serve.
Potential Legislative and Regulatory Responses
The appellate court’s decision is likely to trigger a wave of legislative and regulatory activity. Congress may consider amendments to the 340B program legislation to clarify ambiguities and address the concerns raised by the ruling. This could involve refining the definition of “covered entities” or establishing clearer guidelines on drug manufacturer participation and pricing. Regulatory agencies, such as the Health Resources and Services Administration (HRSA), might issue new guidance or interpretations to provide more concrete direction to covered entities and manufacturers.
We might see increased oversight and enforcement to ensure compliance with revised regulations. For example, Congress could enact legislation specifying that drug manufacturers are required to provide discounts under 340B to contract pharmacies that meet specific criteria, addressing the court’s concerns about the program’s scope.
Potential Areas for Further Legal Challenges
The Novartis case highlights several potential areas for future legal challenges. Disputes may arise over the interpretation of “covered entities” and their eligibility for 340B discounts. The definition of “average manufacturer price” (AMP) and its calculation in relation to 340B pricing could also face further scrutiny. Legal battles might continue over the extent to which manufacturers can restrict or limit their participation in the 340B program.
For instance, future litigation could focus on whether a manufacturer can refuse to provide 340B discounts to contract pharmacies based on the manufacturer’s belief that the pharmacies are not serving the intended patient population. The legal landscape surrounding 340B will likely remain contested for the foreseeable future.
Ongoing Debate Surrounding the Effectiveness and Fairness of the 340B Program
The 340B program’s effectiveness and fairness remain central to the ongoing debate. Supporters argue that it’s crucial for safety-net hospitals and clinics to provide care to vulnerable populations. Critics, however, point to concerns about potential program abuse and the disproportionate impact on drug manufacturers. Some argue that the program leads to inflated drug prices for non-340B patients, while others contend that the savings generated by 340B are vital for maintaining essential healthcare services.
The debate often revolves around the balance between ensuring access to care for underserved communities and maintaining a sustainable pharmaceutical market. For example, some suggest that the program’s success hinges on improved oversight to prevent misuse and ensure that savings are truly used to benefit underserved populations.
Recommendations for Improving the 340B Program
To better serve both patients and healthcare providers, several improvements to the 340B program are needed. These include strengthening oversight and enforcement mechanisms to prevent fraud and abuse. Clearer guidelines and definitions are essential to reduce ambiguity and prevent future legal challenges. Increased transparency regarding 340B pricing and drug manufacturer participation would promote accountability and build public trust.
Furthermore, exploring alternative models for supporting safety-net hospitals and clinics might alleviate some of the concerns raised by drug manufacturers while ensuring continued access to care for vulnerable populations. A thorough review of the program’s overall structure and its impact on various stakeholders is crucial to ensure its long-term sustainability and effectiveness. For example, a tiered system could be implemented to provide greater support to hospitals and clinics serving the most vulnerable populations while providing less support to those with greater financial resources.
Closing Summary
The appellate court’s decision on the 340B program, Novartis, and specialty pharmacies is a significant development with far-reaching consequences. While the ruling offers some clarity, it also raises new questions about the future of the program and its ability to balance the needs of drug manufacturers, healthcare providers, and patients. The ongoing debate underscores the need for a comprehensive review of the 340B program to ensure its effectiveness and fairness in the years to come.
This case isn’t just a legal battle; it’s a reflection of the larger challenges facing our healthcare system in providing access to affordable, life-saving medications.
FAQ Insights
What is the 340B Drug Pricing Program?
It’s a federal program that requires drug manufacturers to provide outpatient drugs at discounted prices to eligible healthcare providers serving a disproportionate number of low-income patients.
How does this ruling affect patients?
The impact on patients is complex and depends on the specific implementation of the ruling. It could potentially affect the availability and affordability of certain medications.
What are the potential long-term effects of this decision?
The long-term effects are uncertain, but they could include changes in drug pricing, shifts in healthcare provider practices, and potential legislative responses.
What is the role of specialty pharmacies in this case?
Specialty pharmacies often dispense high-cost medications, making them a key player in the 340B program and subject to its regulations and potential for disputes.





