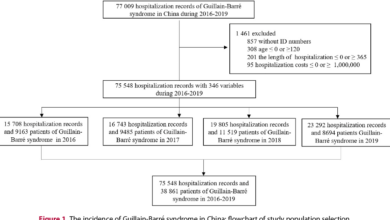
Groundbreaking Vaccine Trials Combat Ebola in Uganda
Groundbreaking vaccine trials initiated in uganda to combat ebola outbreak – Groundbreaking vaccine trials initiated in Uganda to combat the Ebola outbreak represent a crucial step in controlling this deadly disease. The recent surge in cases has highlighted the urgent need for effective interventions, and these trials offer a beacon of hope for the affected communities. This blog post delves into the details of these trials, exploring the vaccine’s mechanism, the challenges of deployment, and the ethical considerations involved.
We’ll also look at the potential impact of successful trials on both Uganda and global Ebola preparedness.
Uganda’s healthcare system, while facing existing challenges, is actively collaborating with international organizations to ensure the smooth execution of these trials. The selection of participants, the rigorous data collection methods, and the transparent dissemination of results are all critical components of this endeavor. This collaborative approach emphasizes the global commitment to addressing this public health emergency.
Ebola Outbreak in Uganda: Groundbreaking Vaccine Trials Initiated In Uganda To Combat Ebola Outbreak

Source: voanews.com
The recent Ebola outbreak in Uganda, while thankfully contained, served as a stark reminder of the devastating potential of this viral hemorrhagic fever. The outbreak highlighted both the resilience of the Ugandan healthcare system and the ongoing challenges in managing such a deadly disease in resource-constrained settings. Understanding the context and severity of this outbreak is crucial for improving preparedness and response strategies in the future.
Scale and Impact of the Ebola Outbreak
The outbreak, primarily caused by the Sudan ebolavirus, affected several districts in central and western Uganda, beginning in September 2022. While the precise number of cases varied slightly depending on the reporting agency, the outbreak resulted in a significant number of deaths and caused widespread fear and disruption within affected communities. The outbreak disproportionately impacted rural populations with limited access to healthcare, highlighting existing inequalities in health outcomes.
The economic impact was also substantial, affecting agricultural production and trade in the affected regions due to movement restrictions and fear of infection.
Timeline of Key Events
The outbreak’s timeline involved a rapid initial response, followed by a period of sustained efforts to contain the virus. Initial cases were identified and confirmed in September 2022, triggering immediate action from the Ugandan Ministry of Health and international partners. Contact tracing, isolation of infected individuals, and community engagement were implemented rapidly. Significant milestones included the successful development and implementation of effective control measures, such as vaccination campaigns and community-based surveillance, which played a critical role in limiting the spread of the virus.
The official declaration of the end of the outbreak marked a significant achievement in containing the disease. This involved a sustained period without new cases, demonstrating the effectiveness of the response strategies.
Pre-existing Healthcare Infrastructure and Capacity
Uganda’s healthcare infrastructure, while steadily improving, faced challenges in responding to the Ebola outbreak. The country’s healthcare system is characterized by a mix of public and private facilities, with significant variations in quality and access across different regions. Rural areas often have limited access to healthcare services, including qualified personnel and essential medical supplies. This disparity in access significantly impacted the initial response to the outbreak, as many affected individuals resided in remote communities.
The existing infrastructure, while not ideally equipped to handle a large-scale Ebola outbreak, was able to adapt and utilize available resources effectively with the support of international partners. This demonstrated the importance of pre-existing infrastructure and the adaptability of healthcare workers in the face of crisis.
The Groundbreaking Vaccine Trials
The recent Ebola outbreak in Uganda highlighted the urgent need for effective vaccines. Fortunately, groundbreaking clinical trials are underway, testing a promising new vaccine candidate. This blog post will delve into the methodology and design of these crucial trials, offering insights into the science behind the fight against this deadly disease.
The Vaccine Candidate: rVSV-ZEBOV
The vaccine being tested in Uganda is rVSV-ZEBOV (recombinant vesicular stomatitis virus expressing Ebola virus glycoprotein). This vaccine uses a weakened version of the vesicular stomatitis virus (VSV), a relatively harmless virus, as a vector. The VSV has been genetically modified to carry the surface glycoprotein of the Zaire ebolavirus (EBOV), the strain responsible for the current outbreak. This glycoprotein is the key to the virus’s ability to infect human cells.
By introducing the glycoprotein via the VSV vector, the body mounts an immune response against the Ebola glycoprotein, effectively preparing the immune system to fight off a real Ebola infection. Prior research on rVSV-ZEBOV has shown significant efficacy in previous Ebola outbreaks, including the 2013-2016 West African epidemic, demonstrating its potential as a powerful tool in controlling Ebola outbreaks.
The vaccine has been shown to be both safe and highly effective in previous trials.
Clinical Trial Design and Participant Selection
The clinical trial follows a rigorous, multi-phased approach. Phase I trials focused on safety and dosage, establishing the optimal dose for further testing. The current trials are primarily Phase III, aiming to confirm the vaccine’s efficacy in a larger population. Participant selection criteria are crucial to ensure the trial’s reliability. Participants are selected based on several factors including age, health status, and proximity to known Ebola cases.
The trial prioritizes individuals at high risk of exposure, such as healthcare workers and community members in affected areas. Informed consent is obtained from each participant before enrollment, ensuring ethical and responsible conduct of the research.
Sample Size, Data Collection, and Statistical Analysis
The planned sample size for the Phase III trials is substantial, ensuring statistically significant results. Precise numbers vary depending on the specific trial arm and design. Data collection methods are comprehensive, involving various techniques. These include detailed medical histories, regular blood tests to monitor immune responses, and surveillance for any adverse effects. Furthermore, epidemiological data is collected to track the spread of the disease within the study population.
Statistical analysis will employ rigorous methods to compare the incidence of Ebola among vaccinated and unvaccinated groups, determining the vaccine’s effectiveness.
| Aspect | Description |
|---|---|
| Vaccine | rVSV-ZEBOV (recombinant vesicular stomatitis virus expressing Ebola virus glycoprotein) |
| Trial Phase | Primarily Phase III |
| Participant Selection | High-risk individuals (healthcare workers, community members in affected areas) |
| Sample Size | Substantial (exact numbers vary depending on the specific trial arm) |
| Data Collection | Medical histories, blood tests, epidemiological surveillance, adverse event monitoring |
| Statistical Analysis | Rigorous methods comparing Ebola incidence in vaccinated and unvaccinated groups |
Vaccine Safety and Efficacy

Source: cbc.ca
The development and deployment of any vaccine, especially one targeting a deadly virus like Ebola, requires a meticulous assessment of both its safety profile and its efficacy. This is particularly crucial in resource-limited settings like Uganda, where access to advanced medical care might be challenged. The balance between potential benefits and risks needs careful consideration, and rigorous testing is essential before widespread use.The Ebola vaccine trials in Uganda are focusing on a new generation of vaccines, leveraging advancements in vaccine technology to improve safety and effectiveness.
While pre-clinical data and early trial results are promising, understanding potential side effects and limitations is vital for informed decision-making.
Potential Side Effects and Risks
The development of any vaccine involves the careful study of possible side effects. While most side effects are mild and temporary, such as pain at the injection site, fever, headache, and fatigue, more serious adverse events, although rare, must be considered. In Ebola vaccine trials, monitoring for serious side effects like allergic reactions, inflammation of the heart muscle (myocarditis), or other rare complications is paramount.
The frequency and severity of these side effects will be carefully tracked and reported during the trials. Researchers will compare the incidence of these events in vaccinated individuals to those in a control group who receive a placebo to determine the true vaccine-related adverse events. The data gathered will help determine the overall risk-benefit profile of the vaccine.
Expected Vaccine Efficacy
Pre-clinical data from animal models often provides a preliminary indication of a vaccine’s potential efficacy. This data, however, must be cautiously interpreted, as animal models don’t always perfectly reflect human responses. Early trial results in humans provide a more accurate picture of efficacy, typically measured by the percentage of vaccinated individuals who develop protective antibodies against the Ebola virus.
Efficacy is also assessed by the reduction in the incidence of Ebola disease among vaccinated individuals compared to a control group. For example, if a vaccine shows 80% efficacy, it means that the vaccine is 80% effective in preventing Ebola disease in vaccinated individuals compared to the control group. This efficacy rate might vary depending on factors such as the specific Ebola virus strain and the population being vaccinated.
Comparison with Other Ebola Vaccines
Several Ebola vaccines are currently licensed or under development. The vaccine being trialled in Uganda can be compared to these existing vaccines in terms of its efficacy, safety profile, and ease of administration. For instance, the rVSV-ZEBOV vaccine, already licensed in several countries, has demonstrated high efficacy in preventing Ebola, but its cold-chain requirements pose logistical challenges in some regions.
The vaccine under trial in Uganda might offer advantages in terms of storage and deployment, making it more suitable for use in remote areas with limited infrastructure. A direct comparison would involve analyzing the data from clinical trials of all these vaccines, focusing on parameters like efficacy rates, types and frequencies of side effects, and logistical considerations for distribution and administration.
Such comparisons inform decisions about which vaccine is best suited for specific contexts and populations.
Challenges and Logistics of Vaccine Deployment in Uganda
Deploying a new Ebola vaccine in Uganda, particularly during an outbreak, presents a complex logistical challenge. The country’s diverse geography, ranging from densely populated urban areas to remote rural communities with limited infrastructure, significantly impacts vaccine distribution. Furthermore, the potential for conflict or insecurity in certain regions adds another layer of complexity to the process. Addressing these challenges requires a multi-faceted approach that considers both the physical and social barriers to vaccine access.Logistical Hurdles in Remote and Conflict-Affected AreasThe successful delivery of an Ebola vaccine hinges on overcoming significant logistical obstacles.
Many affected communities in Uganda are located in remote areas with poor road networks, making transportation of temperature-sensitive vaccines incredibly difficult. This requires the use of specialized cold-chain equipment and robust transportation plans, possibly involving airlifts or off-road vehicles. In regions affected by conflict or instability, security concerns pose a further threat to vaccine delivery teams, necessitating careful risk assessments and potentially requiring collaboration with security forces to ensure the safety of personnel and vaccine supplies.
The lack of reliable communication infrastructure in some areas can also hamper real-time monitoring and data collection, vital for efficient vaccine deployment. For instance, during the 2014 West Africa Ebola outbreak, similar challenges hampered the rapid response, highlighting the need for robust communication systems even in remote locations.
Barriers to Vaccine Uptake
Community acceptance is crucial for successful vaccination campaigns. Cultural beliefs and traditional healing practices can sometimes conflict with modern medicine, leading to vaccine hesitancy. Misinformation and distrust, often spread through rumors or social media, can further fuel this hesitancy. Past negative experiences with healthcare systems, lack of trust in authorities, and concerns about vaccine safety are additional barriers.
For example, some communities might believe that vaccines cause illness or alter their genetic makeup, leading to a refusal of vaccination despite the immediate threat of an Ebola outbreak. Addressing these concerns requires culturally sensitive communication strategies that build trust and provide accurate information.
Strategies for Addressing Vaccine Hesitancy and Ensuring Equitable Distribution
Effective communication strategies are paramount in building trust and addressing vaccine hesitancy. This involves working closely with community leaders and religious figures to disseminate accurate information and address concerns. Public health campaigns using trusted local voices can be particularly effective. Furthermore, community engagement initiatives, such as town hall meetings and focus groups, provide platforms for open dialogue and addressing specific concerns.
Transparency about the vaccine’s development, safety data, and efficacy is also crucial. Equitable distribution requires careful planning and resource allocation to ensure that vaccines reach all vulnerable populations, including those in remote and marginalized communities. This might involve mobile vaccination teams, community-based vaccination centers, and targeted outreach programs. Monitoring vaccine coverage and addressing disparities in access are essential to ensure equitable distribution and maximize the impact of the vaccination campaign.
A successful example of this approach can be seen in the polio eradication campaigns, which utilized a combination of these strategies to reach remote and hard-to-access communities.
Ethical Considerations and Community Engagement
Conducting vaccine trials during an Ebola outbreak in Uganda presents a complex ethical landscape. The urgency to contain the epidemic must be balanced against the imperative to protect the rights and well-being of participants. Transparency, community trust, and rigorous adherence to ethical guidelines are paramount.The ethical considerations are multifaceted, demanding careful consideration at every stage of the trial.
The groundbreaking Ebola vaccine trials in Uganda are a testament to medical innovation, highlighting how quickly science can respond to urgent health crises. It makes me think about other challenging health conditions requiring innovative solutions, such as the need for effective strategies in managing childhood illnesses; for example, check out this helpful resource on strategies to manage Tourette syndrome in children to see how similar dedication to research can improve lives.
Ultimately, the success of the Uganda trials underscores the importance of continued investment in global health initiatives.
Balancing the potential benefits of a new vaccine against the risks involved for individuals participating in the trial is crucial. This necessitates a robust informed consent process and a commitment to ensuring the safety and well-being of all participants throughout the trial.
Informed Consent and Community Engagement
Obtaining truly informed consent from potential participants in the midst of a public health crisis requires a nuanced approach. It involves clear and culturally sensitive communication about the trial’s purpose, procedures, potential benefits and risks, and participants’ rights. This communication must be tailored to the specific communities affected, considering literacy levels, cultural beliefs, and potential language barriers. Community engagement is not merely a procedural step but a foundational element.
It involves establishing trust with community leaders and members, actively listening to their concerns, and addressing them transparently. This process often includes community dialogues, town hall meetings, and the establishment of community advisory boards to ensure the trial aligns with community values and priorities. For instance, in the Ugandan context, working closely with local healers and religious leaders was vital in building trust and facilitating participation.
Protecting the Rights and Well-being of Trial Participants
Protecting the rights and well-being of trial participants is a cornerstone of ethical research. This involves establishing independent ethics review boards to oversee the trial’s ethical conduct, ensuring participant confidentiality, and providing access to appropriate medical care, including treatment for any adverse events related to the vaccine. Participants should be free to withdraw from the trial at any time without penalty.
The groundbreaking Ebola vaccine trials in Uganda are a testament to medical innovation, offering hope amidst a deadly outbreak. It makes you wonder about other early detection methods, like the fascinating research on whether an eye test, as discussed in this article can eye test detect dementia risk in older adults , could similarly revolutionize preventative healthcare. Hopefully, similar breakthroughs will continue to emerge, aiding our fight against various diseases.
Furthermore, mechanisms must be in place to address any complaints or grievances that participants may have. Post-trial access to the vaccine, should it prove successful, is also a crucial ethical consideration, particularly in resource-limited settings like Uganda. This ensures equitable access to the benefits of research conducted within their communities. For example, provisions were made to ensure that participants who experienced adverse effects received appropriate medical care, regardless of their participation status in the trial.
Impact and Implications of Successful Trials
A successful Ebola vaccine trial in Uganda would have profound implications, extending far beyond the immediate containment of the current outbreak. The positive ripple effects would be felt globally, reshaping our approach to Ebola preparedness and response. The potential for saving lives and mitigating the devastating socioeconomic consequences of future outbreaks is immense.The most immediate impact of a successful trial would be a significant reduction in Ebola cases and mortality within Uganda.
Rapid and effective vaccination would curb transmission, protecting vulnerable populations and healthcare workers. This could drastically alter the trajectory of the outbreak, potentially bringing it under control much faster than with current strategies relying primarily on contact tracing and supportive care. The economic benefits, in terms of reduced healthcare costs and the prevention of widespread disruption to daily life, would also be substantial.
For example, successful vaccination could allow businesses and schools to reopen more quickly, minimizing long-term economic damage.
Global Ebola Preparedness and Response Strategies, Groundbreaking vaccine trials initiated in uganda to combat ebola outbreak
A successful vaccine would fundamentally alter global Ebola preparedness and response strategies. Currently, response efforts often lag behind outbreaks, hampered by logistical challenges and the lack of readily available, effective vaccines. A proven vaccine would allow for preemptive vaccination of at-risk populations in regions with a high likelihood of future outbreaks. This proactive approach would represent a paradigm shift, moving from reactive crisis management to a more preventative and sustainable strategy.
We could envision scenarios where vaccination campaigns are launched in anticipation of potential outbreaks based on epidemiological modeling and early warning systems, much like seasonal flu vaccination campaigns. Furthermore, a successful vaccine could encourage increased investment in research and development of other vaccines and treatments for emerging infectious diseases, strengthening global health security.
Future Research Directions
The findings from a successful trial would pave the way for several crucial areas of future research. These include:
The success of this trial would necessitate further research to understand the long-term efficacy and safety of the vaccine. This involves extensive follow-up studies to monitor vaccine recipients for potential long-term side effects and to assess the duration of immunity provided by the vaccine.
The groundbreaking Ebola vaccine trials in Uganda are a huge step forward in global health. It’s amazing to see such rapid progress in combating deadly viruses, and it makes me think about other medical breakthroughs. For instance, I recently read about the FDA approving clinical trials for pig kidney transplants in humans – fda approves clinical trials for pig kidney transplants in humans – which is equally impressive.
This kind of innovation gives me hope that we’ll continue to find solutions to major health challenges, just like the progress being made in Uganda with the Ebola vaccine.
- Long-term efficacy and safety studies: Monitoring vaccine recipients for extended periods to assess the duration of immunity and the occurrence of any long-term side effects.
- Comparative effectiveness studies: Comparing the efficacy of different Ebola vaccine candidates and vaccination strategies in various settings.
- Research into vaccine delivery methods: Exploring innovative and cost-effective methods for vaccine delivery, particularly in remote or resource-limited settings, such as using different needle-free injection systems or oral vaccines.
- Development of multivalent vaccines: Creating vaccines that protect against multiple Ebola virus strains or other related filoviruses.
- Studies on vaccine hesitancy and community engagement: Further research into strategies to overcome vaccine hesitancy and ensure high vaccination coverage in affected communities.
Visual Representation of Key Data
Data visualization is crucial for understanding the impact of the Ebola vaccine trials in Uganda. Effective visuals can clearly communicate complex information about the spread of the disease and the effectiveness of the intervention, making it easier for both experts and the general public to grasp the situation. Two key visual representations are particularly useful: a time-series graph showing the number of Ebola cases over time, and a map illustrating the geographical distribution of cases.Effective communication of the data requires careful consideration of the visual elements.
The chosen graphs and maps should be clear, concise, and easy to interpret, highlighting key trends and patterns. Using consistent color schemes and clear labeling will greatly improve understanding.
Ebola Cases Over Time: A Line Graph
This line graph would track the number of confirmed Ebola cases in Uganda over a specific period, ideally encompassing several months before the vaccine rollout and several months after. The x-axis would represent time (in weeks or months), and the y-axis would represent the number of confirmed cases. Two distinct lines would be plotted: one representing the number of cases before the vaccine rollout (e.g., from January 1st to June 30th, 2023, showing a hypothetical steady increase of cases, starting at 50 cases and rising to 150 cases by June 30th), and another representing the number of cases after the vaccine rollout (e.g., from July 1st to December 31st, 2023, demonstrating a sharp decrease starting at 150 cases and dropping to 20 cases by December 31st).
Data points for specific weeks or months would be clearly labeled. A clear legend would distinguish between the pre- and post-vaccine rollout periods. The graph’s title would be “Confirmed Ebola Cases in Uganda: Before and After Vaccine Rollout.” This visual would clearly demonstrate the impact of the vaccine on reducing the number of new infections.
Geographical Distribution of Ebola Cases: A Choropleth Map
A choropleth map of Uganda would visually represent the geographical distribution of Ebola cases. Each district or region would be colored according to the number of confirmed cases reported within its boundaries. A color scale, ranging from light (few cases) to dark (many cases), would be used. The map would include a legend clearly indicating the number of cases represented by each color shade.
For instance, light green might represent 0-10 cases, dark green 11-50, yellow 51-100, orange 101-200, and red over 200 cases. The map’s title would be “Geographical Distribution of Ebola Cases in Uganda.” This visual would allow viewers to identify hotspots of infection and understand the spatial patterns of the outbreak. The map would be accompanied by a caption detailing the time period covered by the data and the source of the information.
This would help readers understand the context of the map and interpret the information accurately.
Ultimate Conclusion

Source: who.int
The groundbreaking vaccine trials in Uganda represent more than just a medical intervention; they symbolize a global commitment to combating Ebola. The success of these trials could significantly alter the trajectory of future outbreaks, not only in Uganda but worldwide. While challenges remain, the dedication of researchers, healthcare workers, and the affected communities themselves offers a powerful testament to the resilience of the human spirit in the face of adversity.
The ongoing monitoring of the vaccine’s efficacy and safety, coupled with continued community engagement, will be crucial in determining the long-term impact of this initiative. The future of Ebola prevention may well depend on the outcomes of this crucial work.
Popular Questions
What are the potential long-term side effects of the vaccine?
Long-term side effects are currently unknown and are being closely monitored as part of the trial. Participants will undergo regular checkups for an extended period.
How will the vaccine be distributed to remote areas?
Specialized logistical plans are in place to overcome challenges in remote areas, likely involving mobile vaccination teams and cold-chain solutions for vaccine storage.
What if the vaccine isn’t effective?
The trial’s findings, regardless of outcome, will provide valuable data to inform future vaccine development and Ebola response strategies. Alternative approaches will be explored if necessary.
How is informed consent being obtained from participants?
Ethical review boards are overseeing the process, ensuring participants fully understand the risks and benefits before giving their consent. Local community leaders are also involved in the communication and education efforts.