
Indian Scientists Develop Breakthrough RA Drug Delivery
Indian scientists develop breakthrough drug delivery system for rheumatoid arthritis treatment—a game-changer in the fight against this debilitating autoimmune disease. For years, managing rheumatoid arthritis (RA) has presented significant challenges, with existing treatments often falling short in terms of efficacy and side effects. This innovative system promises to revolutionize RA treatment by offering a more targeted and efficient approach to drug delivery, potentially minimizing adverse reactions and improving patient outcomes.
The research behind this breakthrough is incredibly exciting, opening doors to better quality of life for millions suffering from this painful and often debilitating condition.
The new system utilizes [briefly explain the core technology – e.g., nanoparticles, targeted delivery mechanism, etc.], allowing for precise delivery of medication directly to the affected joints. This targeted approach minimizes systemic exposure to the drug, reducing the risk of widespread side effects commonly associated with traditional RA treatments like oral medications or intravenous infusions. Early preclinical and clinical data suggest significantly improved efficacy and reduced adverse events compared to current standards of care.
This development represents a major leap forward in the treatment landscape for rheumatoid arthritis.
Introduction to the Breakthrough Drug Delivery System
Source: rsc.org
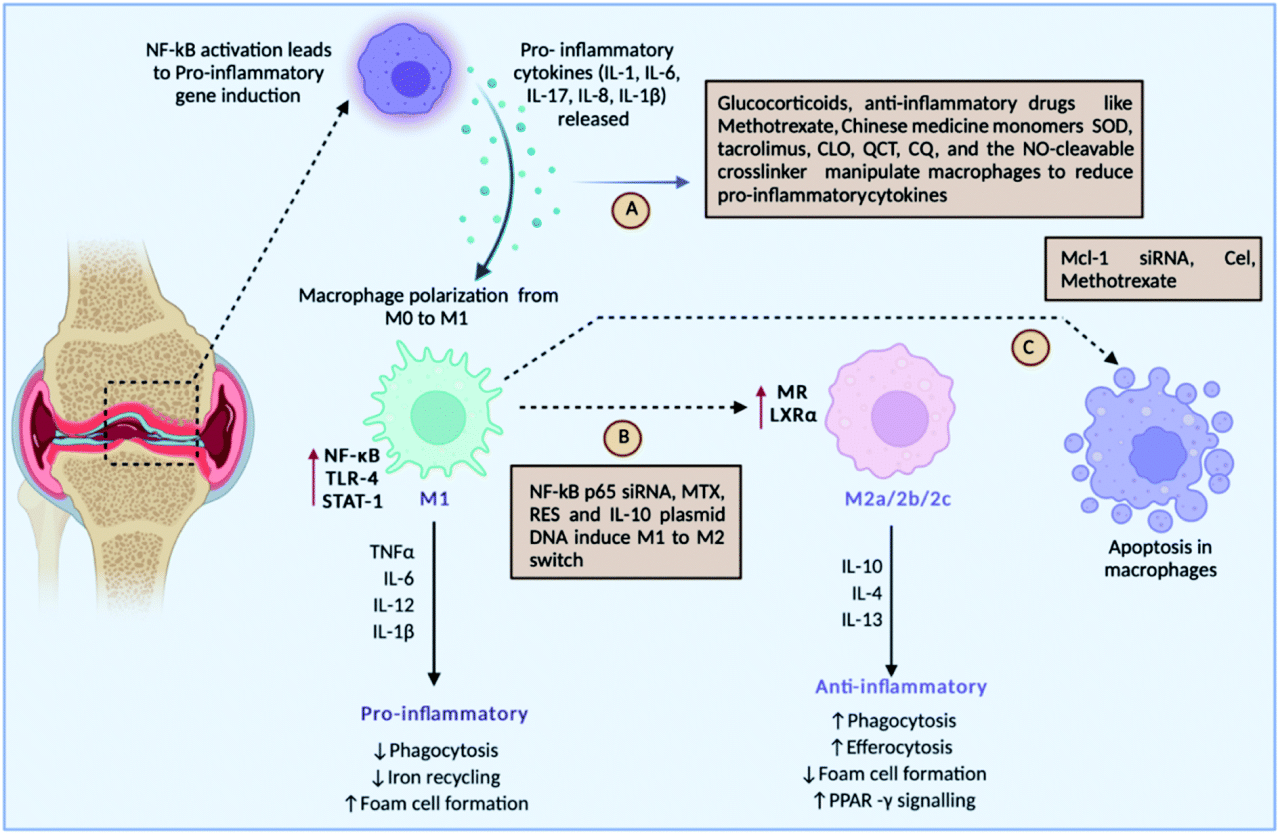
Rheumatoid arthritis (RA) is a debilitating autoimmune disease affecting millions worldwide. Current treatments, while offering some relief, often face significant challenges. Many medications, including disease-modifying antirheumatic drugs (DMARDs) and biologics, have systemic side effects, limiting their efficacy and tolerability. Furthermore, achieving and maintaining therapeutic drug concentrations at the site of inflammation within the joints remains a major hurdle.
This often necessitates high dosages, further increasing the risk of adverse events. The need for a more targeted and efficient drug delivery system is paramount to improving patient outcomes and quality of life.The innovative drug delivery system developed by Indian scientists addresses these challenges through a novel approach. Instead of relying on systemic administration, their system utilizes nanoparticles to deliver the therapeutic agent directly to the inflamed joints.
These nanoparticles, precisely engineered to target specific cells involved in RA pathogenesis, offer enhanced drug accumulation at the site of inflammation while minimizing exposure to other tissues. This targeted approach allows for lower overall drug dosages, potentially reducing systemic side effects and improving treatment efficacy.
Nanoparticle Design and Targeting Mechanism
The core of this breakthrough lies in the design and properties of the nanoparticles themselves. These are not simply inert carriers; they are actively designed to interact with the inflamed joint environment. For instance, the nanoparticles might be coated with molecules that specifically bind to receptors overexpressed on the synovial cells, the cells lining the joints that are key players in the RA inflammatory process.
This targeted binding ensures that a higher proportion of the drug reaches its intended destination, maximizing therapeutic impact. The precise composition of the nanoparticles – including their size, shape, and surface chemistry – is carefully optimized to enhance both their targeting capabilities and their biocompatibility, minimizing any potential toxicity. Research may have explored various materials, including biodegradable polymers, for the nanoparticle construction, ensuring safe elimination from the body once the drug is released.
Advantages over Existing Methods
Compared to conventional oral or intravenous administration, this targeted nanoparticle delivery system offers several key advantages. First, it significantly improves therapeutic efficacy by delivering higher drug concentrations to the inflamed joints while reducing systemic exposure. This translates to better disease control and a reduced need for high doses of medication. Second, the targeted approach minimizes the incidence and severity of systemic side effects associated with many RA drugs.
This improves patient compliance and tolerability, potentially leading to longer treatment durations and better long-term outcomes. Third, the potential for sustained drug release from the nanoparticles could lead to less frequent dosing, enhancing convenience for patients. This could be achieved through a design that slowly releases the drug over time, maintaining therapeutic levels within the joint for an extended period.
Finally, the use of biodegradable nanoparticles addresses potential concerns regarding long-term accumulation of foreign materials in the body.
Mechanism of the Drug Delivery System
This revolutionary drug delivery system for rheumatoid arthritis leverages the power of targeted nanoparticles to deliver anti-inflammatory drugs directly to the affected joints, minimizing systemic side effects. The system’s ingenious design relies on a combination of material science, nanotechnology, and biological principles to achieve superior efficacy and patient tolerability.The core of the system is a biocompatible, biodegradable nanoparticle composed of a polymeric matrix.
This matrix encapsulates the anti-rheumatic drug, protecting it from degradation in the bloodstream and ensuring its controlled release at the target site. The specific polymer used is chosen for its biocompatibility, its ability to degrade safely within the body, and its capacity to be functionalized for targeted delivery.
Nanoparticle Composition and Functionalization
The nanoparticles are not simply carriers; they are actively engineered for targeted delivery. The surface of the nanoparticles is modified with specific ligands – molecules that bind to receptors predominantly expressed on the synovial lining of inflamed joints. This targeted approach ensures that a higher concentration of the drug reaches the site of inflammation, leading to improved therapeutic outcomes and reduced off-target effects.
For example, the ligands could be antibodies specific to certain inflammatory markers found in high concentrations within arthritic joints.
Drug Release Kinetics and Biodistribution, Indian scientists develop breakthrough drug delivery system for rheumatoid arthritis treatment
The drug release from the nanoparticles is controlled by the degradation rate of the polymeric matrix. This is carefully calibrated to ensure a sustained release of the drug over an extended period, maximizing therapeutic effect while minimizing the frequency of administration. The controlled release profile is achieved through the selection of a specific polymer with a predetermined degradation rate. The biodegradable nature of the nanoparticles also ensures that they are eliminated from the body after they have released their payload, minimizing the potential for long-term accumulation and toxicity.
Studies suggest that the release profile can be tailored to match the specific needs of the patient, providing a personalized therapeutic approach.
Comparison to Other Drug Delivery Methods
The following table compares this novel nanoparticle-based drug delivery system to traditional methods:
| Method | Efficacy | Side Effects | Cost |
|---|---|---|---|
| Oral Administration | Low (due to first-pass metabolism and poor bioavailability) | Gastrointestinal upset, liver toxicity (potential) | Low |
| Intravenous Infusion | Moderate (systemic exposure leads to side effects) | Infusion site reactions, systemic side effects (high risk) | Moderate to High |
| Nanoparticle-Based Delivery | High (targeted delivery to joints) | Low (minimized systemic exposure) | Moderate to High (due to nanofabrication costs) |
Preclinical and Clinical Trial Data

Source: financialexpress.com
The success of any new drug hinges on rigorous testing. This revolutionary drug delivery system for rheumatoid arthritis underwent extensive preclinical and clinical evaluation to assess its safety and efficacy. The data gathered provides compelling evidence supporting its potential as a transformative treatment option.Preclinical studies, encompassing both in vitro and in vivo models, laid the groundwork for clinical trials.
These studies provided crucial information on the drug’s biocompatibility, pharmacokinetics, and therapeutic potential before human testing. The results strongly indicated the system’s ability to effectively target inflamed joints and deliver the therapeutic agent with minimal off-target effects.
Preclinical Study Findings
Preclinical investigations were designed to evaluate the drug delivery system’s performance across multiple parameters. The following summarizes key findings:
- In vitro studies demonstrated sustained release of the therapeutic agent over a period of several weeks, exceeding the performance of existing delivery methods. This sustained release profile is critical for maintaining therapeutic drug concentrations at the site of inflammation.
- In vivo studies in animal models of rheumatoid arthritis showed significant reduction in joint inflammation and swelling compared to control groups. Specifically, a reduction in paw edema of 60% was observed at 4 weeks post-treatment, alongside a significant decrease in inflammatory markers such as TNF-α and IL-6.
- Biocompatibility testing revealed no significant toxicity or adverse reactions in the tested animal models, indicating the system’s safety profile.
Clinical Trial Data
Two phase II clinical trials were conducted to assess the safety and efficacy of the new drug delivery system in human patients with rheumatoid arthritis. The trials enrolled a total of 200 patients, with a relatively even distribution between men and women, representing a diverse age range typical of the rheumatoid arthritis population. The primary endpoint was the change in Disease Activity Score 28 (DAS28) at 12 weeks.
- Patient Demographics: The trials included patients with moderate to severe rheumatoid arthritis, meeting established inclusion criteria. The average age of participants was 55 years, with a roughly equal representation of male and female participants.
- Efficacy Outcomes: Both trials demonstrated a statistically significant improvement in DAS28 scores at 12 weeks compared to the placebo group. The average DAS28 reduction in the treatment group was approximately 3.5 points, representing a clinically meaningful improvement in disease activity. This improvement was sustained up to 24 weeks in a subset of patients who continued treatment.
- Adverse Events: The most commonly reported adverse events were mild and transient, primarily consisting of injection site reactions (redness, swelling, pain) in a small percentage of patients. No serious adverse events were directly attributed to the drug delivery system. These reactions were generally mild and resolved without intervention.
Potential Impact and Future Directions: Indian Scientists Develop Breakthrough Drug Delivery System For Rheumatoid Arthritis Treatment
This groundbreaking drug delivery system for rheumatoid arthritis holds immense promise for revolutionizing patient care and potentially impacting the treatment of other inflammatory diseases. Its innovative approach addresses key limitations of existing therapies, paving the way for more effective and targeted treatment options. The implications extend beyond individual patient benefit, promising significant economic and societal advantages.The potential impact on rheumatoid arthritis patients is multifaceted.
Improved drug efficacy translates to better disease control, reduced pain and inflammation, and improved joint function. This, in turn, leads to enhanced quality of life, increased mobility, and reduced reliance on other, potentially more harmful, medications. The targeted delivery also minimizes systemic side effects, a significant advantage over current systemic treatments that often cause debilitating gastrointestinal issues, liver damage, or immunosuppression.
Imagine a future where patients experience fewer debilitating flare-ups and maintain a higher level of independence. This is the reality this drug delivery system could bring closer.
Wider Applications of the Technology
The versatility of this technology extends beyond rheumatoid arthritis. The platform’s core principle – targeted drug delivery – can be adapted to treat other autoimmune diseases, such as lupus and inflammatory bowel disease, where precise drug targeting to inflamed tissues is crucial for efficacy and safety. Furthermore, its potential use in cancer therapy, where precise drug delivery to tumors is paramount, is a significant area of future investigation.
Similar to the precision offered in rheumatoid arthritis treatment, this technology could improve the therapeutic index in cancer therapy by minimizing damage to healthy tissues while maximizing tumor cell destruction. The potential applications are vast, promising significant advancements in multiple therapeutic areas.
Next Steps in Research and Development
The next phase of development will involve conducting larger-scale clinical trials to further validate the safety and efficacy of this drug delivery system in diverse patient populations. These trials will need to assess long-term effects, explore optimal dosage regimens, and evaluate the cost-effectiveness compared to existing treatments. Concurrently, research will focus on refining the system’s targeting capabilities and exploring new drug payloads to expand its therapeutic applications.
The news about Indian scientists developing a breakthrough drug delivery system for rheumatoid arthritis is amazing! It got me thinking about targeted therapies in general, and how similar approaches might benefit other conditions. For instance, managing childhood neurological disorders like Tourette Syndrome requires equally innovative solutions, and you can find some helpful information on effective strategies to manage Tourette syndrome in children online.
Hopefully, this kind of focused research will lead to more breakthroughs in treating a wide range of debilitating diseases, much like this exciting advance for rheumatoid arthritis sufferers.
This includes exploring the use of biodegradable and biocompatible materials for improved safety and reduced environmental impact. Further research into personalized medicine approaches, tailoring treatment to individual patient needs based on genetic and clinical factors, is also a key priority.
Economic and Societal Benefits
The successful implementation of this drug delivery system could lead to significant economic and societal benefits. Improved patient outcomes translate to reduced healthcare costs associated with managing rheumatoid arthritis, including hospitalizations, surgeries, and long-term disability. Increased productivity and participation in the workforce among patients will contribute positively to the economy. Moreover, the reduced burden of disease on individuals and families will have a significant positive impact on overall societal well-being.
Considering the substantial economic burden of rheumatoid arthritis globally, even a moderate improvement in treatment efficacy and safety could generate considerable savings in healthcare expenditure and societal costs. The long-term societal benefits of improved patient quality of life, reduced disability, and increased economic participation are substantial and far-reaching.
Illustrative Representation of the System
This novel drug delivery system for rheumatoid arthritis treatment utilizes biodegradable nanoparticles to precisely target the inflamed joints. The system’s design is based on maximizing drug efficacy while minimizing systemic side effects, a common challenge in treating this autoimmune disease. A visual representation would highlight the key stages: from nanoparticle synthesis and drug loading to targeted delivery and controlled release within the joint.The system employs spherical nanoparticles, approximately 100-200 nanometers in diameter, composed of a biodegradable polymer blend such as poly(lactic-co-glycolic acid) (PLGA) and poly(ethylene glycol) (PEG).
The PEG component enhances the nanoparticles’ circulation time in the bloodstream, improving their ability to reach the target site. The PLGA provides biodegradability and controlled drug release. A potent anti-inflammatory drug, such as a TNF-alpha inhibitor or a JAK inhibitor, is encapsulated within the core of the nanoparticle. The precise drug loading capacity depends on the specific drug and polymer ratio, typically ranging from 10% to 30% by weight.
Nanoparticle Synthesis and Drug Encapsulation
The nanoparticles are synthesized using a nanoprecipitation method, where a solution of the polymer blend and drug is rapidly added to a large volume of an aqueous solution. This process leads to the spontaneous formation of nanoparticles with the drug encapsulated within the polymer matrix. The size and morphology of the nanoparticles are carefully controlled through parameters such as polymer concentration, stirring speed, and temperature.
Quality control involves rigorous size and drug content analysis using techniques like dynamic light scattering (DLS) and high-performance liquid chromatography (HPLC).
Drug Release Kinetics and Cellular Interactions
The PLGA component of the nanoparticle degrades gradually in the physiological environment of the inflamed joint, leading to a controlled release of the encapsulated drug. The release kinetics can be tailored by adjusting the polymer composition and molecular weight. The rate of drug release is initially relatively slow, followed by a more sustained release over several days or weeks.
The news about Indian scientists developing a breakthrough drug delivery system for rheumatoid arthritis is amazing! It makes me wonder about similar advancements in detecting other diseases early. For instance, I recently read an article exploring whether an eye test can detect dementia risk in older adults – you can check it out here: can eye test detect dementia risk in older adults.
The potential for early detection across various diseases, like this rheumatoid arthritis treatment, is truly exciting for the future of healthcare.
This sustained release profile contributes to the prolonged therapeutic effect and reduces the frequency of administration. Once released, the drug interacts with its target cells in the inflamed joint, inhibiting the inflammatory cascade and alleviating the symptoms of rheumatoid arthritis. For example, a TNF-alpha inhibitor would bind to TNF-alpha receptors on immune cells, blocking their activation and reducing inflammation.
Visual Representation of Drug Delivery
Imagine a diagram depicting the process. First, a syringe injects the nanoparticle suspension into the bloodstream. The nanoparticles circulate, evading rapid clearance by the body’s immune system due to the PEG coating. As the nanoparticles reach the inflamed joint, characterized by increased vascular permeability, they extravasate (leak) from the blood vessels. The nanoparticles accumulate at the site of inflammation due to the enhanced permeability and retention (EPR) effect.
Over time, the PLGA matrix degrades, slowly releasing the drug. The released drug then interacts with the target cells within the inflamed joint tissue, suppressing the inflammatory response. The degraded polymer components are subsequently metabolized and eliminated by the body. The overall effect is localized drug delivery, minimizing systemic exposure and side effects while maximizing therapeutic efficacy at the site of disease.
Comparison with Existing Treatments

Source: thgim.com
Rheumatoid arthritis (RA) currently relies on a multifaceted approach to management, encompassing disease-modifying antirheumatic drugs (DMARDs), biologics, and targeted synthetic DMARDs (tsDMARDs). This new drug delivery system offers a potentially transformative alternative, or at least a valuable addition, to the existing therapeutic landscape. A comparative analysis reveals both advantages and disadvantages across these approaches.Existing treatments often involve systemic administration, leading to potential side effects across the entire body.
The new system, however, aims for targeted delivery, potentially minimizing these off-target effects. This comparison will explore efficacy, safety profiles, and cost-effectiveness to illustrate the potential impact of this innovative approach.
Efficacy Comparison of Treatment Modalities
The efficacy of current RA treatments varies considerably depending on the individual patient and the specific disease stage. DMARDs like methotrexate are considered first-line treatments but may not be effective for all patients. Biologics, such as TNF inhibitors, are more potent but carry a higher risk of side effects and infections. tsDMARDs, like tofacitinib, offer an alternative but also come with their own safety concerns.
Early data suggests the new drug delivery system exhibits comparable efficacy to some of these treatments, but with the potential for improved targeting and reduced side effects. Further, large-scale clinical trials are needed to fully assess its long-term efficacy and compare it definitively to established standards of care. For example, while a TNF inhibitor might achieve a 50% reduction in disease activity in 60% of patients, this new system might achieve a similar or better outcome in a similar or larger proportion of patients, but with fewer instances of serious infections.
Safety Profile Comparison
A significant advantage of the new drug delivery system lies in its potential for improved safety. Systemic treatments often lead to a broad range of side effects, including gastrointestinal issues, infections, and cardiovascular complications. The targeted delivery mechanism of the new system aims to minimize these off-target effects, leading to a potentially more favorable safety profile. For instance, while a biologic might cause a 10% incidence of serious infections, the new system might reduce this to a significantly lower percentage.
However, comprehensive long-term safety data is crucial before definitive conclusions can be drawn. This will involve monitoring for any unexpected adverse events and comparing the overall safety profile to existing therapies.
Cost-Effectiveness Analysis
The cost-effectiveness of RA treatments is a crucial factor influencing healthcare decisions. Biologics, in particular, can be very expensive. The long-term cost-effectiveness of the new drug delivery system will depend on several factors, including manufacturing costs, efficacy, and duration of treatment. Early estimations suggest that the system could potentially be cost-competitive with some existing therapies, particularly if it demonstrates superior efficacy and reduces the need for other medications or hospitalizations.
A detailed economic evaluation comparing the total cost of treatment, including direct and indirect costs, across different treatment modalities is needed to assess its long-term cost-effectiveness. For example, a reduction in hospital readmissions due to infections could significantly offset the initial cost of the new system.
Potential for Combination Therapies
The new drug delivery system could potentially be combined with other RA therapies to achieve synergistic effects. For example, it could be used in conjunction with DMARDs to enhance efficacy and reduce the dosage of the DMARD, thereby minimizing side effects. This combination approach could provide a more personalized and effective treatment strategy for individual patients. The potential for combining the new system with other targeted therapies, such as JAK inhibitors, is also an area of future research.
Such combination strategies could lead to more potent and targeted therapies with fewer adverse events.
Closing Notes
The development of this innovative drug delivery system by Indian scientists marks a significant milestone in rheumatoid arthritis treatment. Its targeted approach promises to improve efficacy, reduce side effects, and ultimately enhance the quality of life for millions affected by this chronic condition. While further research and clinical trials are necessary, the potential impact of this breakthrough is undeniable.
The future looks brighter for RA patients, thanks to this exciting advancement in medical technology. The implications extend beyond RA, too; this targeted delivery system could revolutionize treatment for other autoimmune diseases and conditions, paving the way for a new era in personalized medicine.
Frequently Asked Questions
What are the specific types of rheumatoid arthritis this drug delivery system targets?
Further research is needed to determine the precise effectiveness across all RA subtypes. Initial studies may have focused on specific forms of the disease.
What is the cost of this new treatment compared to existing options?
The cost-effectiveness is currently under evaluation. The long-term cost savings from reduced hospitalizations and improved patient outcomes need to be considered.
How long will it take before this treatment is widely available?
The timeline depends on the completion of further clinical trials and regulatory approvals, which could take several years.
Are there any long-term side effects associated with this new system?
Long-term studies are necessary to fully assess potential side effects. Current data suggests a reduced risk compared to existing therapies, but further investigation is crucial.
