Gustave Roussy Pioneers Precision Oncology with DuOnco Unity AI for Comprehensive Tumor Burden Assessment
The landscape of cancer treatment evaluation is undergoing a fundamental transformation as Gustave Roussy, Europe’s leading cancer center, unveils DuOnco Unity, an advanced artificial intelligence solution designed to redefine how clinicians measure therapeutic efficacy. For decades, the global standard for assessing a patient’s response to cancer treatment has been the RECIST (Response Evaluation Criteria in Solid Tumors) framework. While RECIST has provided a necessary degree of standardization, it is increasingly viewed as an incomplete metric in the era of precision medicine. By shifting the focus from a handful of "target lesions" to a comprehensive, three-dimensional assessment of the total tumor burden, DuOnco Unity represents a significant leap forward in oncological imaging and personalized patient care.
The Limitations of Traditional RECIST Criteria
To understand the impact of DuOnco Unity, one must first examine the constraints of the current clinical gold standard. The RECIST criteria, established in the early 2000s and updated in 2009 (RECIST 1.1), rely on the manual measurement of a limited number of lesions—typically no more than five in total, with a maximum of two per organ. These lesions are selected by radiologists to serve as proxies for the patient’s entire disease state. The success or failure of a chemotherapy, immunotherapy, or targeted therapy is then determined by comparing the change in the sum of the longest diameters of these specific targets over time.
However, this reductionist approach faces significant hurdles in complex cases. Many patients with metastatic disease present with dozens or even hundreds of lesions across multiple organs. Selecting five "representative" targets can be subjective and may fail to capture the heterogeneous response of the disease; for instance, some tumors may shrink while others grow or new ones appear elsewhere. Furthermore, RECIST is primarily a one-dimensional measurement (diameter), which does not always correlate accurately with the actual volume of a tumor, particularly those with irregular shapes. In the context of modern oncology, where characterizing the exhaustive extent of a tumor is vital for adapting therapeutic strategies, the need for a more global evaluation of "tumor burden" has become a central priority for the medical community.
A Strategic Academic-Industrial Partnership
The development of DuOnco Unity is the result of a high-level consortium that bridges the gap between clinical research and industrial innovation. Led by Gustave Roussy, the project involves a strategic collaboration with Guerbet, a global leader in medical imaging contrast agents; Intrasense, a specialist in advanced software solutions for medical imaging; and the University Hospital (CHU) of Angers. This multidisciplinary effort was made possible through significant financial backing from BPI France and the "France 2030" investment program, a government initiative aimed at positioning France as a global leader in healthcare innovation and artificial intelligence.
The partnership leverages the unique strengths of each member. Gustave Roussy provides the oncological expertise and the massive datasets required for training; Guerbet and Intrasense contribute the technological infrastructure and algorithmic development; and CHU d’Angers assists in the clinical validation of the tool. This synergy has produced a tool that is not only scientifically robust but also commercially viable and ready for integration into hospital workflows.
Technical Specifications: The Power of 3D AI Analysis
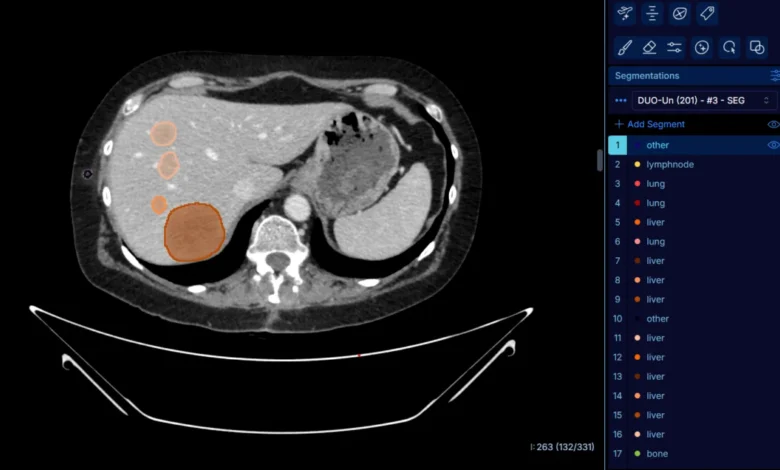
DuOnco Unity is built upon a sophisticated deep-learning algorithm trained on a massive scale. To achieve the necessary accuracy, researchers and radiologists annotated approximately 287,000 individual lesions to train the AI. This extensive dataset allows the algorithm to automatically detect and segment tumors and metastases in three dimensions across thoraco-abdomino-pelvic (TAP) CT scans—the most common imaging modality for monitoring solid cancers.
Unlike existing AI tools that focus on a single organ, DuOnco Unity is multi-organ and multi-anatomical. It analyzes the entire thorax, abdomen, and pelvis in a single pass. One of its most distinguishing features is its ability to identify and measure lymph nodes, which are among the most common sites for metastasis but are notoriously difficult to assess consistently using manual methods.
Beyond tumor metrics, the tool integrates "opportunistic screening" by analyzing muscle mass and adipose tissue (fat). This is a critical addition to oncological reports, as sarcopenia (the loss of muscle mass) is a significant prognostic factor for treatment toxicity and overall survival. By providing a holistic view of the patient’s body composition alongside their tumor burden, DuOnco Unity gives oncologists a much more nuanced understanding of the patient’s physiological resilience.
Efficiency and Integration in Clinical Practice
One of the most immediate benefits of DuOnco Unity is the drastic reduction in the time required for comprehensive imaging analysis. Manually identifying and measuring every single lesion in a patient with widespread metastatic disease is a task so labor-intensive that it is virtually impossible in routine clinical practice. Such a task could take a radiologist hours. DuOnco Unity, however, delivers a complete 3D analysis in just 3 to 15 minutes.
Crucially, the tool has been designed to integrate seamlessly into the existing digital environment of radiology departments. The interface is intuitive, and the system is built for local data hosting to ensure patient privacy and security, avoiding the need to export sensitive medical images to external servers. Furthermore, the AI is "backwards compatible." It can analyze contemporary scans as well as archival images from older machines that lack native AI capabilities. This allows clinicians to re-evaluate a patient’s historical disease progression with a level of detail that was previously unavailable, providing a richer long-term view of the disease’s trajectory.
Clinical Validation: The IMPACT and LIFE Studies
While the technological promise of DuOnco Unity is clear, its clinical utility is currently being rigorously tested through two major studies at Gustave Roussy.
- The IMPACT Study: This trial involves 600 patients suffering from lung cancer and melanoma. The primary objective is to determine whether the AI-detected "total tumor burden" is a more accurate predictor of progression-free survival (PFS) and overall survival (OS) than traditional RECIST measurements. By quantifying the entire volume of the disease, researchers hope to identify subtle changes in tumor mass that could signal treatment failure months earlier than current methods.
- The LIFE Study: This study focuses on the human element of the technology. It evaluates the impact of DuOnco Unity on the daily clinical practice of physicians. Researchers are looking at how the availability of these advanced metrics influences the speed and confidence of medical decision-making, as well as the level of standardization it brings to radiology reports across different practitioners.
Currently, DuOnco Unity is operating as a prototype at Gustave Roussy while it undergoes the necessary steps for CE marking, the regulatory approval required for commercial distribution and use in standard clinical care across Europe.
Implications for Precision Oncology and Patient Outcomes
The shift from 1D proxy measurements to 3D total tumor burden quantification has profound implications for the future of oncology. In the current model, a patient might remain on an ineffective treatment for several months because their "target lesions" have not yet met the RECIST threshold for "progressive disease," even if other non-target areas are worsening. DuOnco Unity allows for the earlier identification of treatment resistance, enabling oncologists to pivot to alternative therapies sooner. Conversely, it can also confirm the efficacy of a treatment that might appear stable on a 1D plane but is actually shrinking in volume.
By minimizing the time patients spend on ineffective therapies, the medical community can reduce unnecessary side effects and improve the quality of life for those living with cancer. Furthermore, the objective and reproducible nature of AI measurements reduces the variability between different radiologists, ensuring that a patient’s evaluation remains consistent regardless of which hospital or specialist performs the scan.
Professor Nathalie Lassau, a radiologist at Gustave Roussy and an international expert in AI-applied radiology, emphasized the transformative nature of this tool. "With DuOnco Unity, we are changing the scale of imaging analysis in oncology," she stated. "We can now understand the disease in its entirety. For radiologists, this means a considerable gain in time and a valuable aid in standardizing the evaluation of tumor burden. For oncologists, it provides much more complete indicators to evaluate treatment effectiveness. This innovation allows us to move from a partial estimate to a global, quantitative, and reproducible vision of the disease."
About Gustave Roussy: A Global Leader in Cancer Innovation
The development of DuOnco Unity reinforces Gustave Roussy’s position as a premier global institution. Ranked as the top cancer center in France and Europe, and sixth worldwide, the institute operates as a comprehensive hub for patient care, research, and teaching. As a founding member of the Paris-Saclay Cancer Cluster, Gustave Roussy treats over 54,000 patients annually, including nearly 3,000 children and adolescents.
With a workforce of 4,000 employees across its Villejuif and Chevilly-Larue sites, the institute is dedicated to treating all types of cancer at all stages of life. Its commitment to innovation is reflected in its clinical trial participation rate, with over 40% of its patients included in clinical studies. The introduction of DuOnco Unity is the latest milestone in the institute’s mission to combine cutting-edge technology with humane, personalized care, ultimately aiming to improve the survival and well-being of cancer patients globally.
As the oncology community awaits the full results of the IMPACT and LIFE studies and the subsequent CE marking of DuOnco Unity, the tool stands as a testament to the power of artificial intelligence to not only assist the physician but to provide a deeper, more accurate window into the biological reality of cancer. This move toward "Total Tumor Burden" assessment may soon become the new international benchmark, ushering in a more precise era of cancer management.








